QUESTION IMAGE
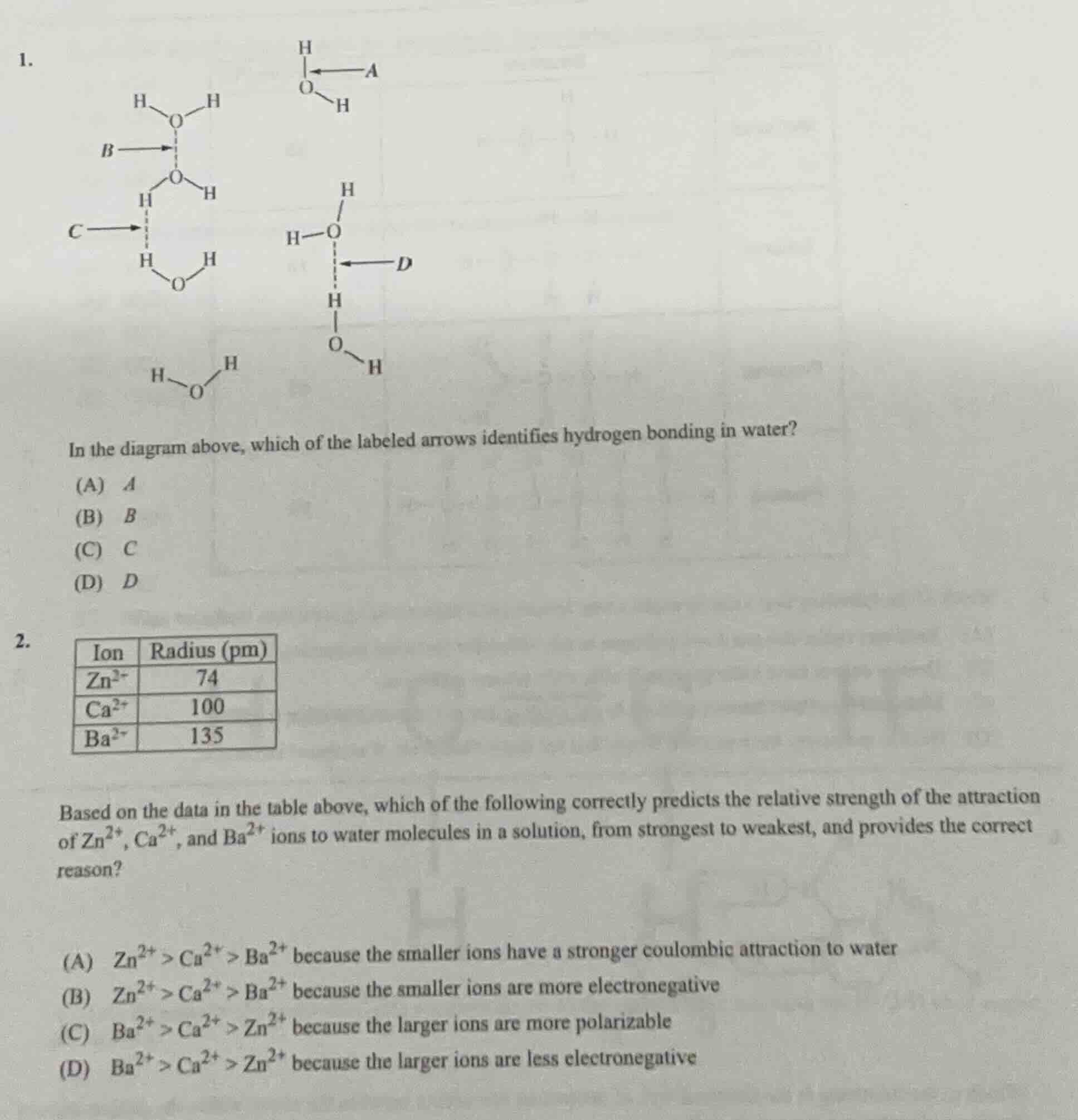
Question
- in the diagram above, which of the labeled arrows identifies hydrogen bonding in water? (a) a (b) b (c) c (d) d 2. | ion | radius (pm) | | ---- | ---- | | zn²⁺ | 74 | | ca²⁺ | 100 | | ba²⁺ | 135 | based on the data in the table above, which of the following correctly predicts the relative strength of the attraction of zn²⁺, ca²⁺, and ba²⁺ ions to water molecules in a solution, from strongest to weakest, and provides the correct reason? (a) zn²⁺ > ca²⁺ > ba²⁺ because the smaller ions have a stronger coulombic attraction to water (b) zn²⁺ > ca²⁺ > ba²⁺ because the smaller ions are more electronegative (c) ba²⁺ > ca²⁺ > zn²⁺ because the larger ions are more polarizable (d) ba²⁺ > ca²⁺ > zn²⁺ because the larger ions are less electronegative
Step1: Analyze hydrogen bonding rules
Hydrogen bonding occurs between a hydrogen atom covalently bonded to a highly electronegative atom (O, N, F) and a lone pair on another highly electronegative atom. Arrow A is an intramolecular O-H bond; Arrow B points to an O atom without a H attached to an electronegative atom for the interaction; Arrow C points to a H not bonded to the electronegative O of the adjacent molecule; Arrow D points from the lone pair of an O atom to a H atom covalently bonded to another O atom, fitting hydrogen bonding.
Step2: Analyze ion-water attraction
Coulombic attraction follows $F \propto \frac{q_1q_2}{r}$, where all ions have +2 charge ($q_1$), water's dipole charge ($q_2$) is constant. Smaller ionic radius ($r$) means stronger attraction. Ionic radii: $\text{Zn}^{2+}$ (74 pm) < $\text{Ca}^{2+}$ (100 pm) < $\text{Ba}^{2+}$ (135 pm), so attraction strength is $\text{Zn}^{2+} > \text{Ca}^{2+} > \text{Ba}^{2+}$, with the reason being smaller ions have stronger coulombic attraction.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- (D) D
- (A) $\text{Zn}^{2+} > \text{Ca}^{2+} > \text{Ba}^{2+}$ because the smaller ions have a stronger coulombic attraction to water