QUESTION IMAGE
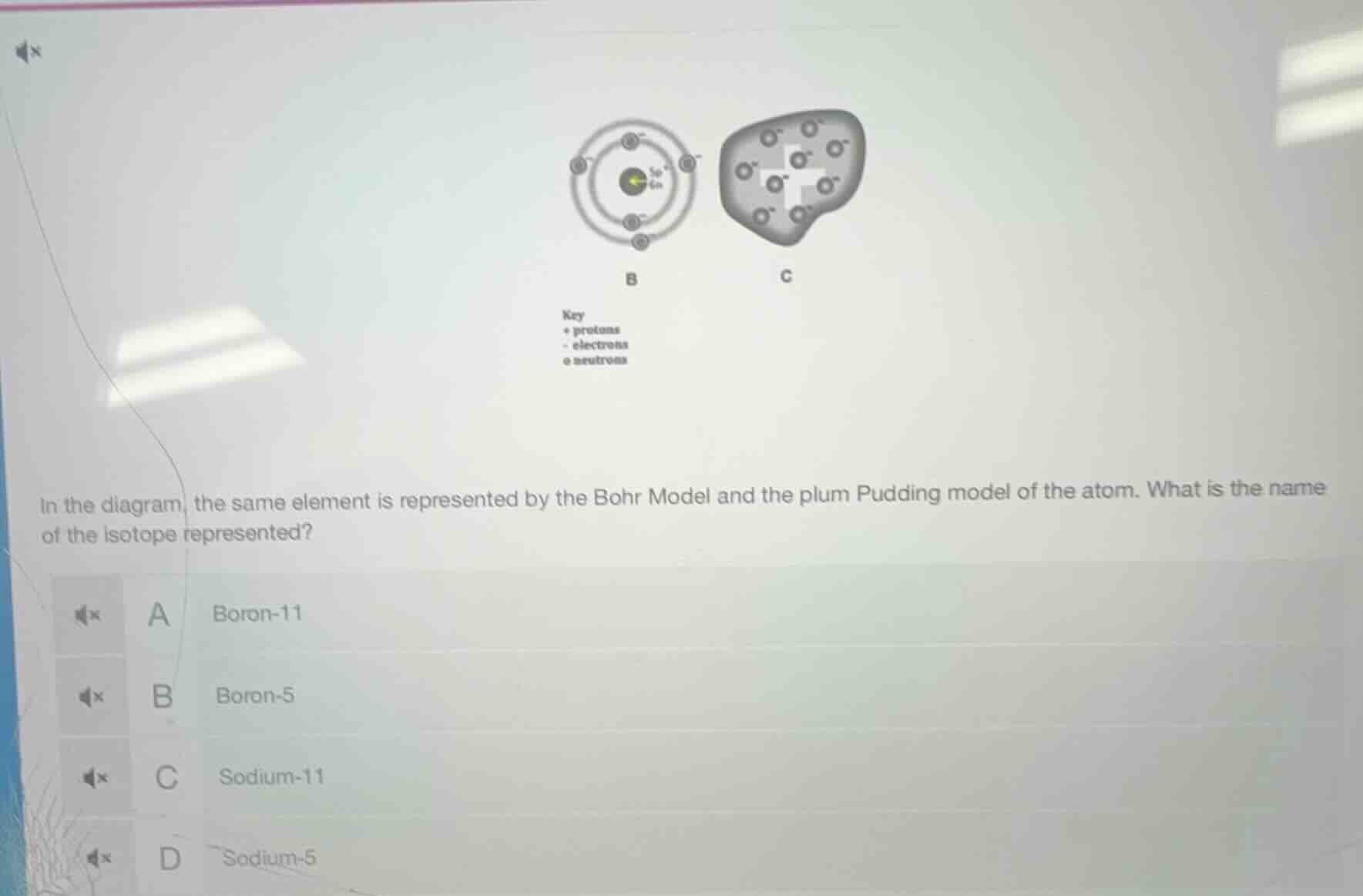
Question
in the diagram, the same element is represented by the bohr model and the plum pudding model of the atom. what is the name of the isotope represented?
a boron-11
b boron-5
c sodium-11
d sodium-5
Step1: Determine the number of protons
From the Bohr model (diagram B), the number of protons (positive charges in the nucleus) is 5 (since the key shows + as protons, and we can count the protons in the nucleus). The number of protons determines the element, so an element with 5 protons is Boron (atomic number 5).
Step2: Determine the number of neutrons
From the diagrams, in the nucleus of the Bohr model, we can see the neutrons (the uncharged particles). Counting them, we have 6 neutrons. The mass number of an isotope is the sum of protons and neutrons, so \( 5 + 6 = 11 \).
Step3: Identify the isotope
Since the element with 5 protons is Boron, and the mass number is 11, the isotope is Boron - 11.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. Boron - 11