QUESTION IMAGE
Question
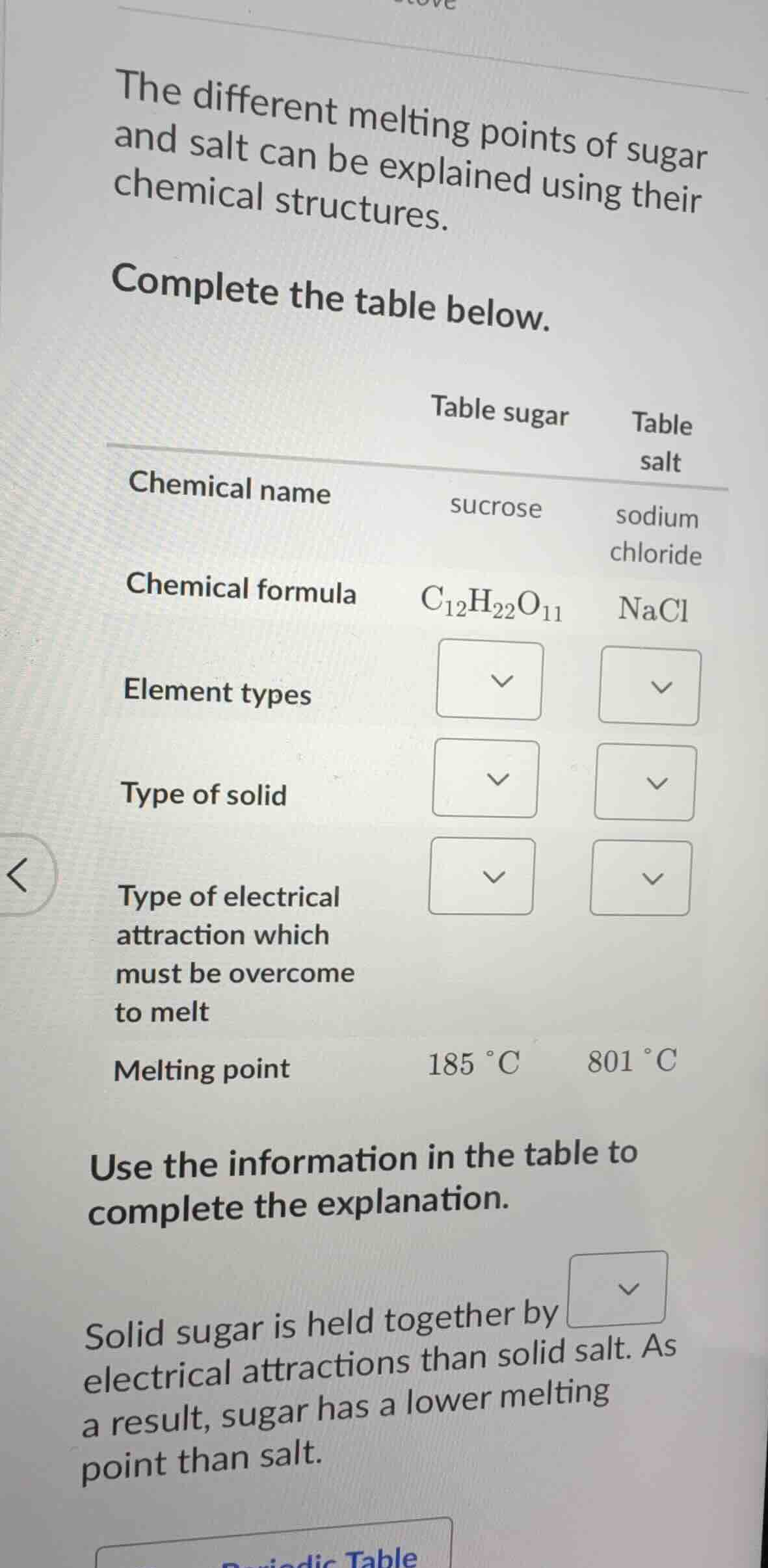
the different melting points of sugar and salt can be explained using their chemical structures. complete the table below. table sugar table salt chemical name sucrose sodium chloride chemical formula $ce{c_{12}h_{22}o_{11}}$ $ce{nacl}$ element types $\boldsymbol{downarrow}$ $\boldsymbol{downarrow}$ type of solid $\boldsymbol{downarrow}$ $\boldsymbol{downarrow}$ type of electrical attraction which must be overcome to melt $\boldsymbol{downarrow}$ $\boldsymbol{downarrow}$ melting point $185 ^{circ}\text{c}$ $801 ^{circ}\text{c}$ use the information in the table to complete the explanation. solid sugar is held together by $\boldsymbol{downarrow}$ electrical attractions than solid salt. as a result, sugar has a lower melting point than salt.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Table sugar - Element types: Carbon, Hydrogen, Oxygen
Table sugar - Type of solid: Molecular solid
Table sugar - Type of electrical attraction which must be overcome to melt: Hydrogen bonds and dipole-dipole forces, London dispersion forces
Table salt - Element types: Sodium, Chlorine
Table salt - Type of solid: Ionic solid
Table salt - Type of electrical attraction which must be overcome to melt: Ionic bonds
Solid sugar is held together by weaker electrical attractions than solid salt. As a result, sugar has a lower melting point than salt.