QUESTION IMAGE
Question
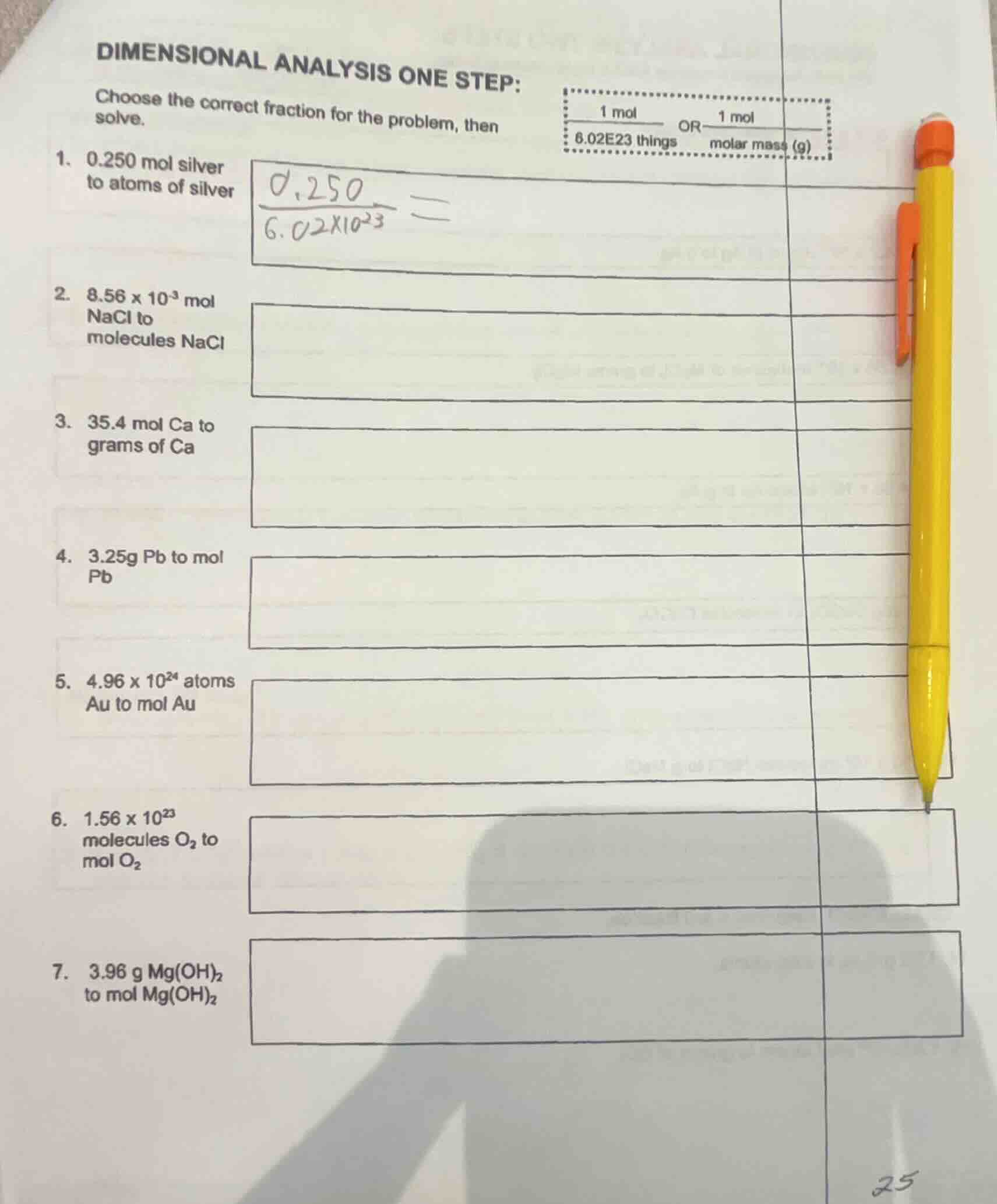
dimensional analysis one step:
choose the correct fraction for the problem, then solve.
$\frac{1 \text{mol}}{6.02e23 \text{things}}$ or $\frac{1 \text{mol}}{\text{molar mass (g)}}$
- 0.250 mol silver to atoms of silver
$\frac{0.250}{6.02×10^{23}} =$
- $8.56 × 10^{-3}$ mol nacl to molecules nacl
- 35.4 mol ca to grams of ca
- 3.25g pb to mol pb
- $4.96 × 10^{24}$ atoms au to mol au
- $1.56 × 10^{23}$ molecules $o_2$ to mol $o_2$
- 3.96 g $mg(oh)_2$ to mol $mg(oh)_2$
25
Step1: Moles to atoms (Avogadro's number)
$0.250\ \text{mol Ag} \times 6.02 \times 10^{23}\ \text{atoms/mol} = 1.51 \times 10^{23}\ \text{atoms Ag}$
Step2: Moles to molecules (Avogadro's number)
$8.56 \times 10^{-3}\ \text{mol NaCl} \times 6.02 \times 10^{23}\ \text{molecules/mol} = 5.15 \times 10^{21}\ \text{molecules NaCl}$
Step3: Moles to grams (molar mass Ca=40.08g/mol)
$35.4\ \text{mol Ca} \times 40.08\ \text{g/mol} = 1419\ \text{g Ca}$
Step4: Grams to moles (molar mass Pb=207.2g/mol)
$3.25\ \text{g Pb} \times \frac{1\ \text{mol}}{207.2\ \text{g}} = 0.0157\ \text{mol Pb}$
Step5: Atoms to moles (Avogadro's number)
$4.96 \times 10^{24}\ \text{atoms Au} \times \frac{1\ \text{mol}}{6.02 \times 10^{23}\ \text{atoms}} = 8.24\ \text{mol Au}$
Step6: Molecules to moles (Avogadro's number)
$1.56 \times 10^{23}\ \text{molecules }O_2 \times \frac{1\ \text{mol}}{6.02 \times 10^{23}\ \text{molecules}} = 0.259\ \text{mol }O_2$
Step7: Grams to moles (molar mass $\text{Mg(OH)}_2=58.32$g/mol)
$3.96\ \text{g Mg(OH)}_2 \times \frac{1\ \text{mol}}{58.32\ \text{g}} = 0.0679\ \text{mol Mg(OH)}_2$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $1.51 \times 10^{23}$ atoms of silver
- $5.15 \times 10^{21}$ molecules of NaCl
- $1419$ grams of Ca
- $0.0157$ mol Pb
- $8.24$ mol Au
- $0.259$ mol $O_2$
- $0.0679$ mol $\text{Mg(OH)}_2$