QUESTION IMAGE
Question
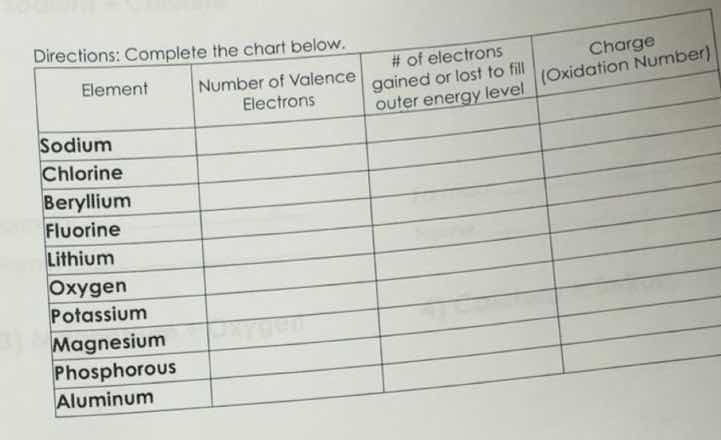
directions: complete the chart below.
| element | number of valence electrons | # of electrons gained or lost to fill outer energy level | charge (oxidation number) |
|---|---|---|---|
| chlorine | |||
| beryllium | |||
| fluorine | |||
| lithium | |||
| oxygen | |||
| potassium | |||
| magnesium | |||
| phosphorous | |||
| aluminum |
Step1: Find valence electrons
Valence electrons = group number (main groups):
- Sodium (Group1): $1$
- Chlorine (Group17): $7$
- Beryllium (Group2): $2$
- Fluorine (Group17): $7$
- Lithium (Group1): $1$
- Oxygen (Group16): $6$
- Potassium (Group1): $1$
- Magnesium (Group2): $2$
- Phosphorus (Group15): $5$
- Aluminum (Group13): $3$
Step2: Determine electron gain/loss
Atoms fill outer level (8 e⁻, except H/He):
- Metals lose electrons; nonmetals gain:
- Sodium: Lose $1$
- Chlorine: Gain $1$
- Beryllium: Lose $2$
- Fluorine: Gain $1$
- Lithium: Lose $1$
- Oxygen: Gain $2$
- Potassium: Lose $1$
- Magnesium: Lose $2$
- Phosphorus: Gain $3$
- Aluminum: Lose $3$
Step3: Calculate oxidation charge
Charge = -(e⁻ gained) or +(e⁻ lost):
- Sodium: $+1$
- Chlorine: $-1$
- Beryllium: $+2$
- Fluorine: $-1$
- Lithium: $+1$
- Oxygen: $-2$
- Potassium: $+1$
- Magnesium: $+2$
- Phosphorus: $-3$
- Aluminum: $+3$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Element | Number of Valence Electrons | # of electrons gained or lost to fill outer energy level | Charge (Oxidation Number) |
|---|---|---|---|
| Chlorine | 7 | Gains 1 | -1 |
| Beryllium | 2 | Loses 2 | +2 |
| Fluorine | 7 | Gains 1 | -1 |
| Lithium | 1 | Loses 1 | +1 |
| Oxygen | 6 | Gains 2 | -2 |
| Potassium | 1 | Loses 1 | +1 |
| Magnesium | 2 | Loses 2 | +2 |
| Phosphorous | 5 | Gains 3 | -3 |
| Aluminum | 3 | Loses 3 | +3 |