QUESTION IMAGE
Question
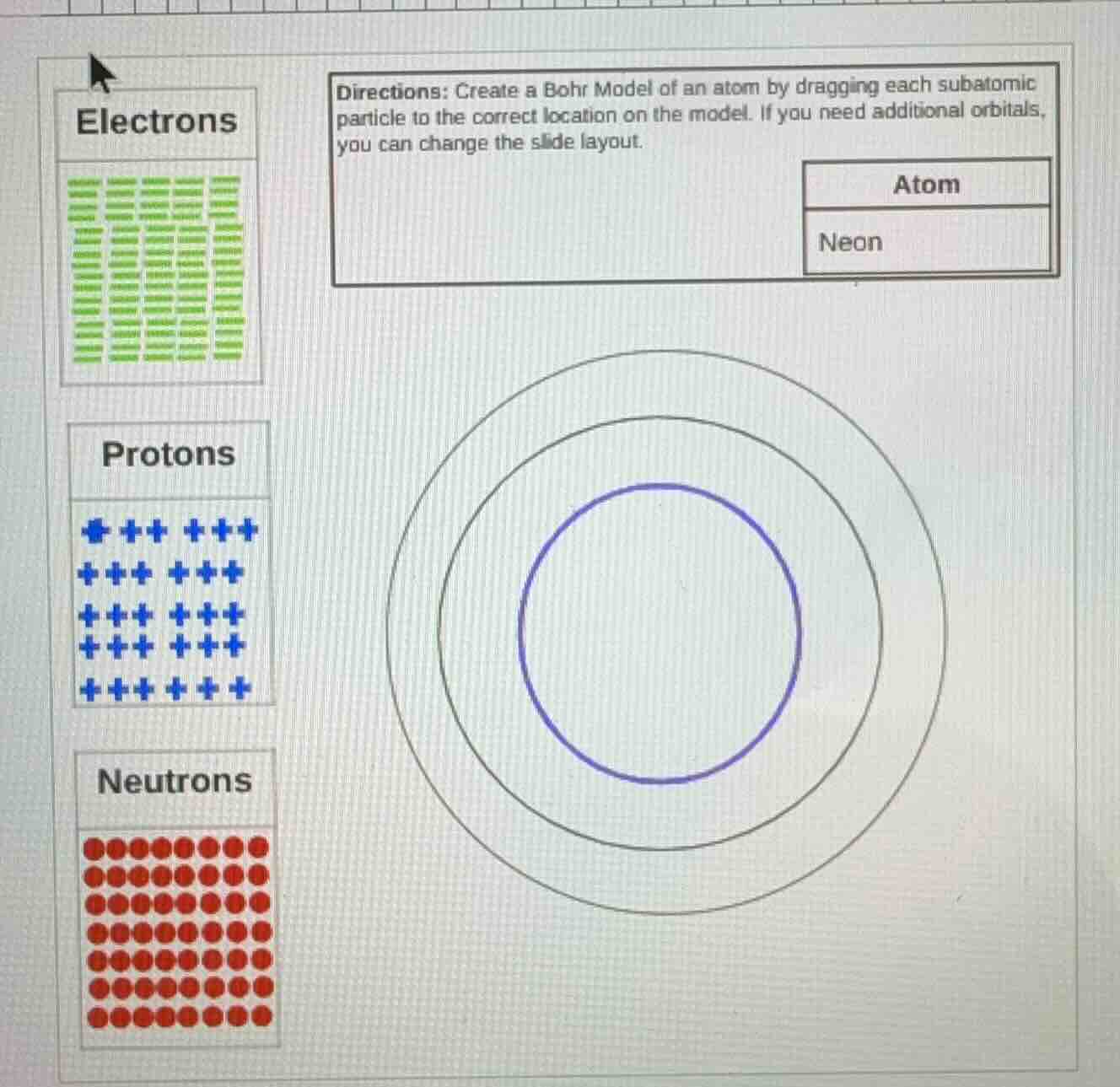
directions: create a bohr model of an atom by dragging each subatomic particle to the correct location on the model. if you need additional orbitals, you can change the slide layout.
atom
neon
electrons
protons
neutrons
Step1: Find Neon's atomic number
Neon (Ne) has an atomic number of 10, so it has 10 protons.
Step2: Determine electron count
Neutral atoms have equal protons/electrons: 10 electrons.
Step3: Find neutron count
Neon's most abundant isotope (Ne-20) has a mass number of 20. Neutrons = mass number - protons = $20 - 10 = 10$.
Step4: Assign particles to model
- Place 10 protons + 10 neutrons in the central blue circle (nucleus).
- The first electron shell holds max 2 electrons: place 2 electrons on the innermost ring.
- The second electron shell holds max 8 electrons: place 8 electrons on the middle ring (outer ring for neon, as it only needs 2 shells).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Nucleus (central blue circle): 10 blue protons + 10 red neutrons
- Innermost electron ring: 2 green electrons
- Middle (outer) electron ring: 8 green electrons