QUESTION IMAGE
Question
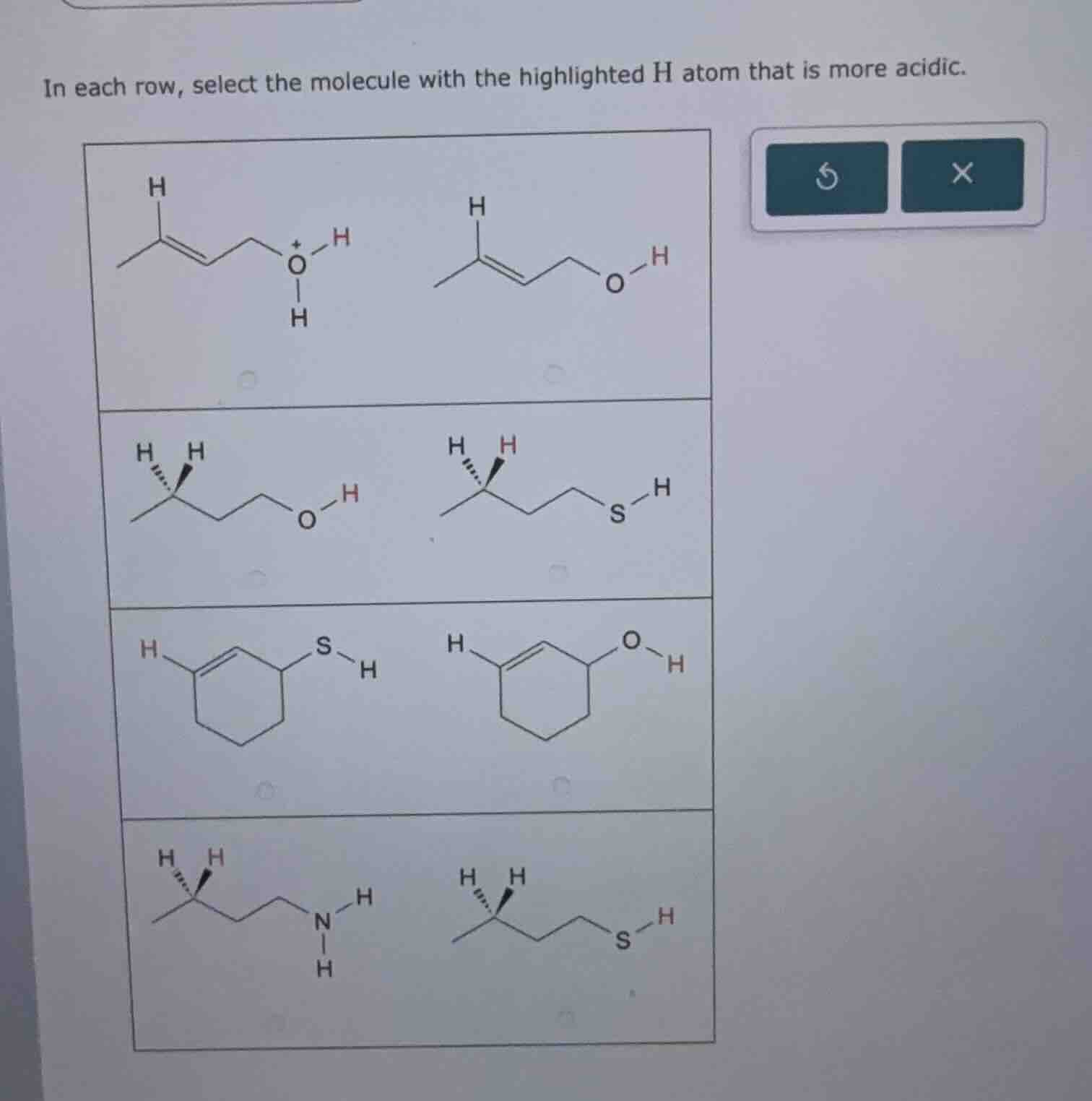
in each row, select the molecule with the highlighted h atom that is more acidic.
Row 1:
Step1: Analyze the groups
The first molecule has a protonated hydroxyl group ($\ce{-O^+H_2}$) and the second has a neutral hydroxyl group ($\ce{-OH}$). A protonated oxygen (in $\ce{-O^+H_2}$) is more electron - withdrawing, making its $\ce{H}$ more acidic.
Step2: Choose the more acidic
So the first molecule (with $\ce{O^+ - H}$) has a more acidic highlighted $\ce{H}$.
Row 2:
Step1: Compare $\ce{O - H}$ and $\ce{S - H}$
Sulfur is larger than oxygen, so the $\ce{S - H}$ bond is longer and weaker. A weaker bond means the $\ce{H}$ is more easily dissociated (more acidic). The first molecule has $\ce{O - H}$, the second has $\ce{S - H}$.
Step2: Choose the more acidic
So the second molecule (with $\ce{S - H}$) has a more acidic highlighted $\ce{H}$.
Row 3:
Step1: Compare $\ce{S - H}$ and $\ce{O - H}$
As sulfur is larger than oxygen, the $\ce{S - H}$ bond is weaker than the $\ce{O - H}$ bond. A weaker bond implies a more acidic $\ce{H}$. The first molecule has $\ce{S - H}$, the second has $\ce{O - H}$.
Step2: Choose the more acidic
So the first molecule (with $\ce{S - H}$) has a more acidic highlighted $\ce{H}$.
Row 4:
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s:
Row 1: The first molecule (with $\ce{O^+ - H}$)
Row 2: The second molecule (with $\ce{S - H}$)
Row 3: The first molecule (with $\ce{S - H}$)
Row 4: The second molecule (with $\ce{S - H}$)