QUESTION IMAGE
Question
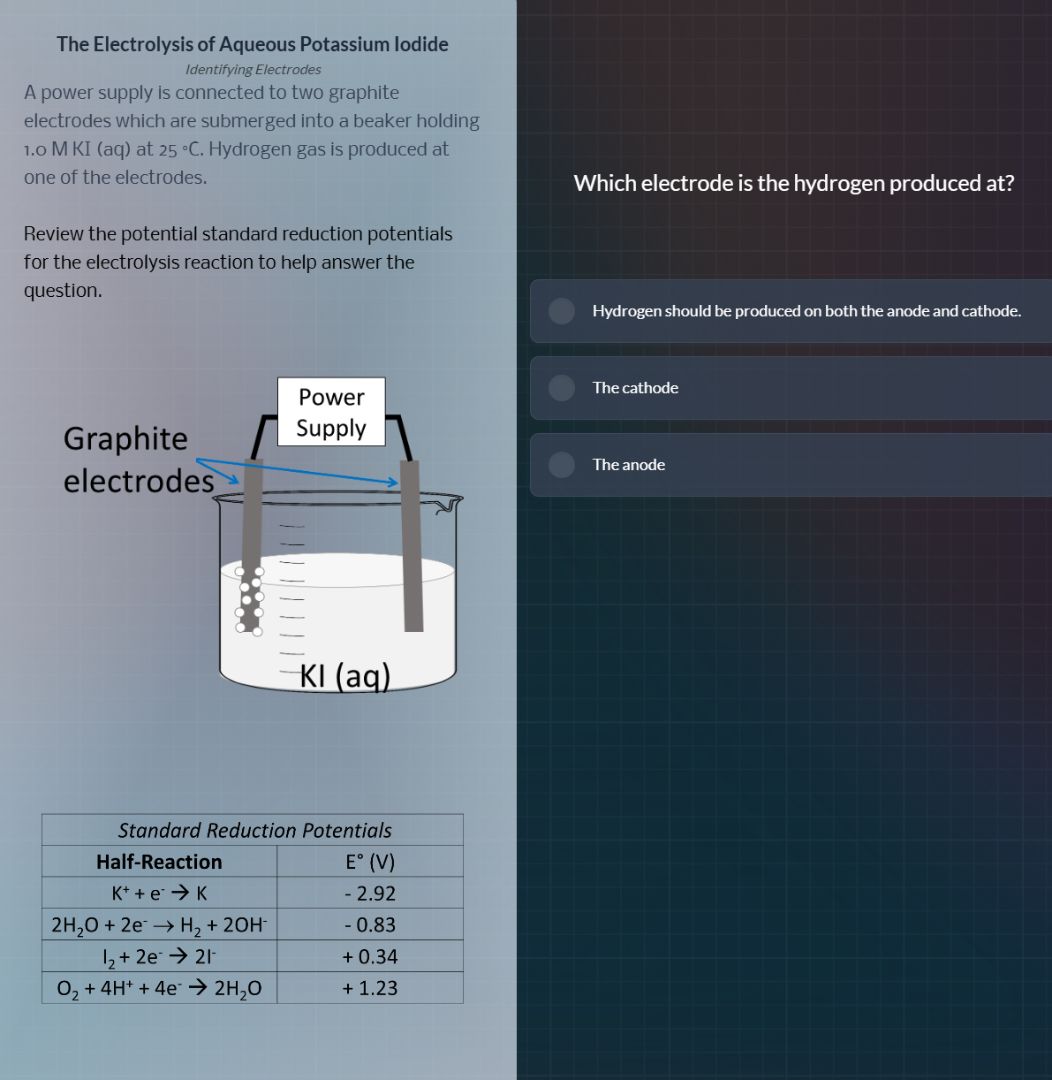
the electrolysis of aqueous potassium iodide
identifying electrodes
a power supply is connected to two graphite
electrodes which are submerged into a beaker holding
1.0 m ki (aq) at 25 °c. hydrogen gas is produced at
one of the electrodes.
review the potential standard reduction potentials
for the electrolysis reaction to help answer the
question.
which electrode is the hydrogen produced at?
hydrogen should be produced on both the anode and cathode.
the cathode
the anode
standard reduction potentials
half - reaction e° (v)
k⁺ + e⁻ → k - 2.92
2h₂o + 2e⁻ → h₂ + 2oh⁻ - 0.83
i₂ + 2e⁻ → 2i⁻ + 0.34
o₂ + 4h⁺ + 4e⁻ → 2h₂o + 1.23
In electrolysis, reduction occurs at the cathode (gain of electrons) and oxidation at the anode (loss of electrons). For aqueous KI, possible reduction half - reactions are \(K^{+}+e^{-}\to K\) (\(E^{\circ}=- 2.92\ V\)) and \(2H_{2}O + 2e^{-}\to H_{2}+2OH^{-}\) (\(E^{\circ}=-0.83\ V\)). The half - reaction with the less negative (more positive) standard reduction potential is more likely to occur. Since \(-0.83\ V\) (for water reduction to \(H_{2}\)) is more positive than \(-2.92\ V\) (for \(K^{+}\) reduction), the reduction of water to produce \(H_{2}\) occurs at the cathode.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The cathode