QUESTION IMAGE
Question
electrons and the periodic table
quiz complete
1 2 3 4 5 6 7 8 9 10
time remaining
59:58
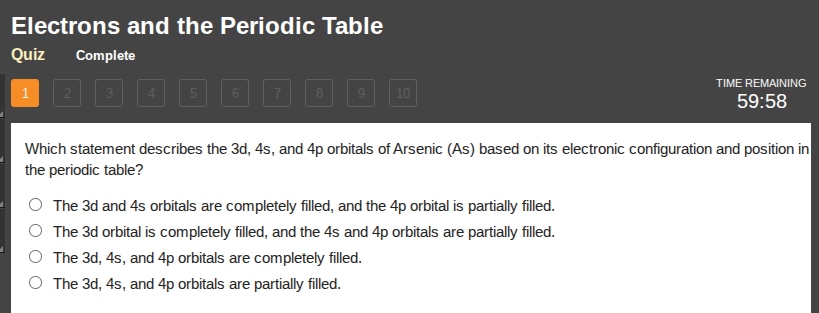
which statement describes the 3d, 4s, and 4p orbitals of arsenic (as) based on its electronic configuration and position in the periodic table?
the 3d and 4s orbitals are completely filled, and the 4p orbital is partially filled.
the 3d orbital is completely filled, and the 4s and 4p orbitals are partially filled.
the 3d, 4s, and 4p orbitals are completely filled.
the 3d, 4s, and 4p orbitals are partially filled.
$$1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^{10} 4p^3$$
Step3: Analyze orbital filling
- 4s: $4s^2$ (full, holds max 2)
- 3d: $3d^{10}$ (full, holds max 10)
- 4p: $4p^3$ (partial, holds max 6)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$$1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^{10} 4p^3$$
Step3: Analyze orbital filling
- 4s: $4s^2$ (full, holds max 2)
- 3d: $3d^{10}$ (full, holds max 10)
- 4p: $4p^3$ (partial, holds max 6)