QUESTION IMAGE
Question
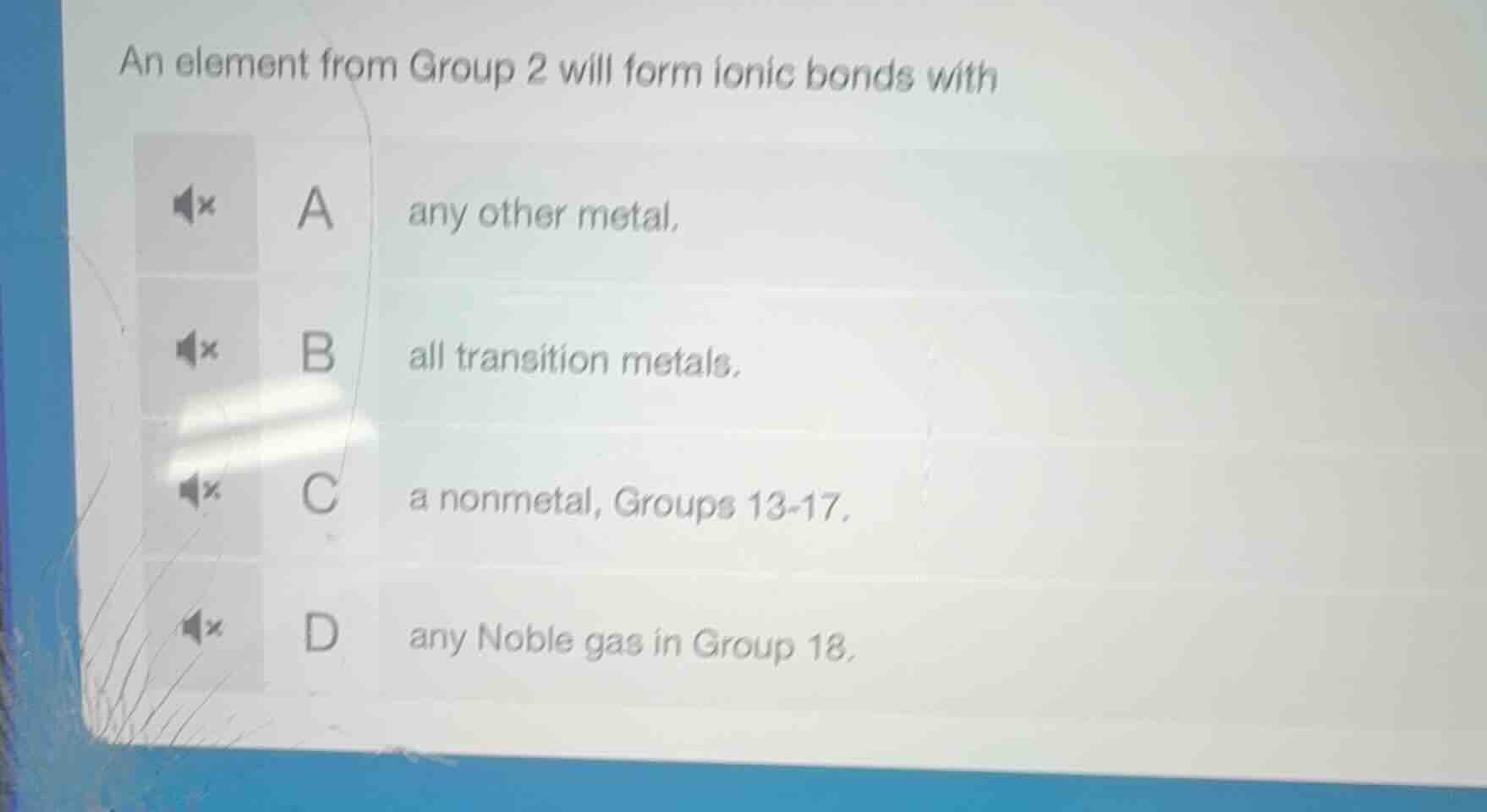
an element from group 2 will form ionic bonds with
a any other metal.
b all transition metals.
c a nonmetal, groups 13-17.
d any noble gas in group 18.
Group 2 elements are metals (alkaline earth metals) that tend to lose 2 electrons to form cations. Ionic bonds form between metals and nonmetals, where nonmetals (usually from Groups 13 - 17, as they can gain electrons to form anions) and metals transfer electrons. Metals don't form ionic bonds with other metals (they form metallic bonds), transition metals are also metals, and noble gases are stable and don't form bonds easily. So a Group 2 element (metal) will form ionic bonds with a nonmetal from Groups 13 - 17.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. a nonmetal, Groups 13 - 17.