QUESTION IMAGE
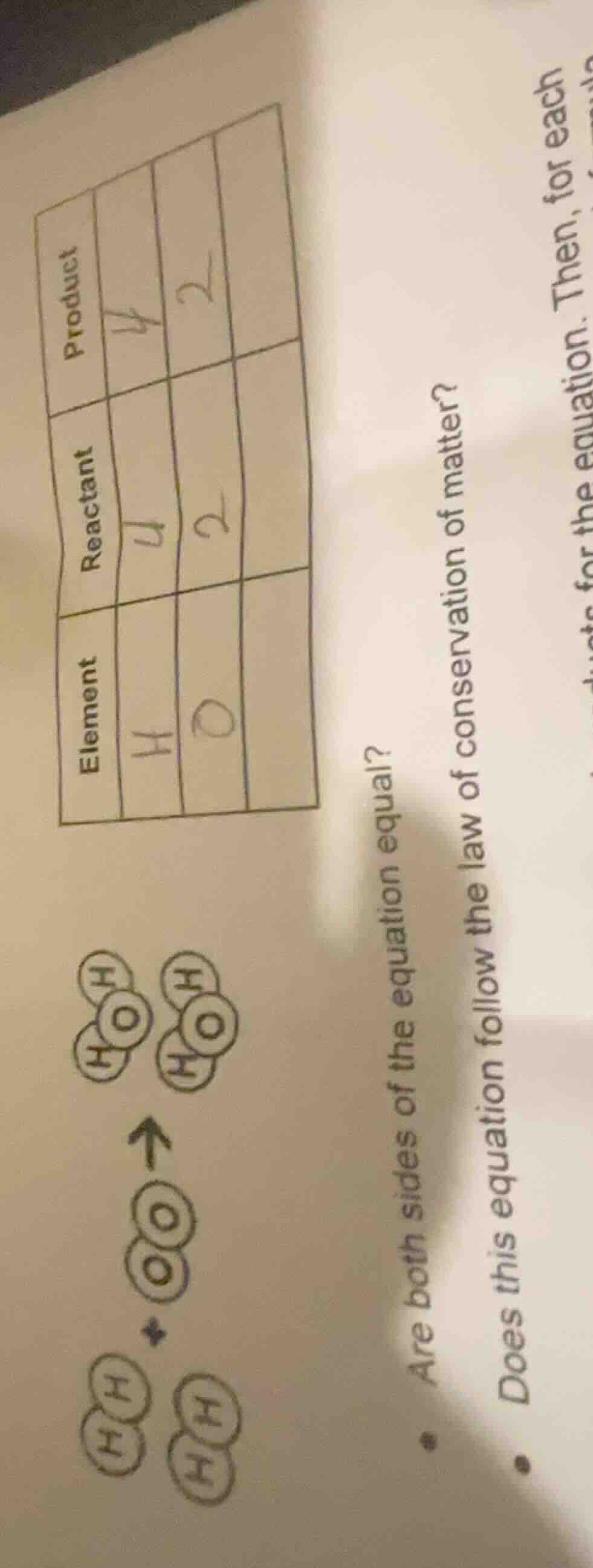
Question
element | reactant | product
h | 4 | 4
o | 2 | 2
are both sides of the equation equal?
does this equation follow the law of conservation of matter?
Step1: Count H atoms (reactants)
Reactants: $2\mathrm{H_2}$ → $2 \times 2 = 4$ H atoms
Step2: Count O atoms (reactants)
Reactants: $\mathrm{O_2}$ → $2$ O atoms
Step3: Count H atoms (products)
Products: $2\mathrm{H_2O}$ → $2 \times 2 = 4$ H atoms
Step4: Count O atoms (products)
Products: $2\mathrm{H_2O}$ → $2 \times 1 = 2$ O atoms
Step5: Compare reactant/product counts
For H: $4 = 4$; For O: $2 = 2$
Step6: Evaluate conservation of matter
Law requires equal atom counts on both sides.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Yes, both sides of the equation are equal (4 H atoms and 2 O atoms on each side).
- Yes, this equation follows the law of conservation of matter, as the number of each type of atom is conserved between reactants and products.
The balanced chemical equation is: $2\mathrm{H_2} + \mathrm{O_2}
ightarrow 2\mathrm{H_2O}$