QUESTION IMAGE
Question
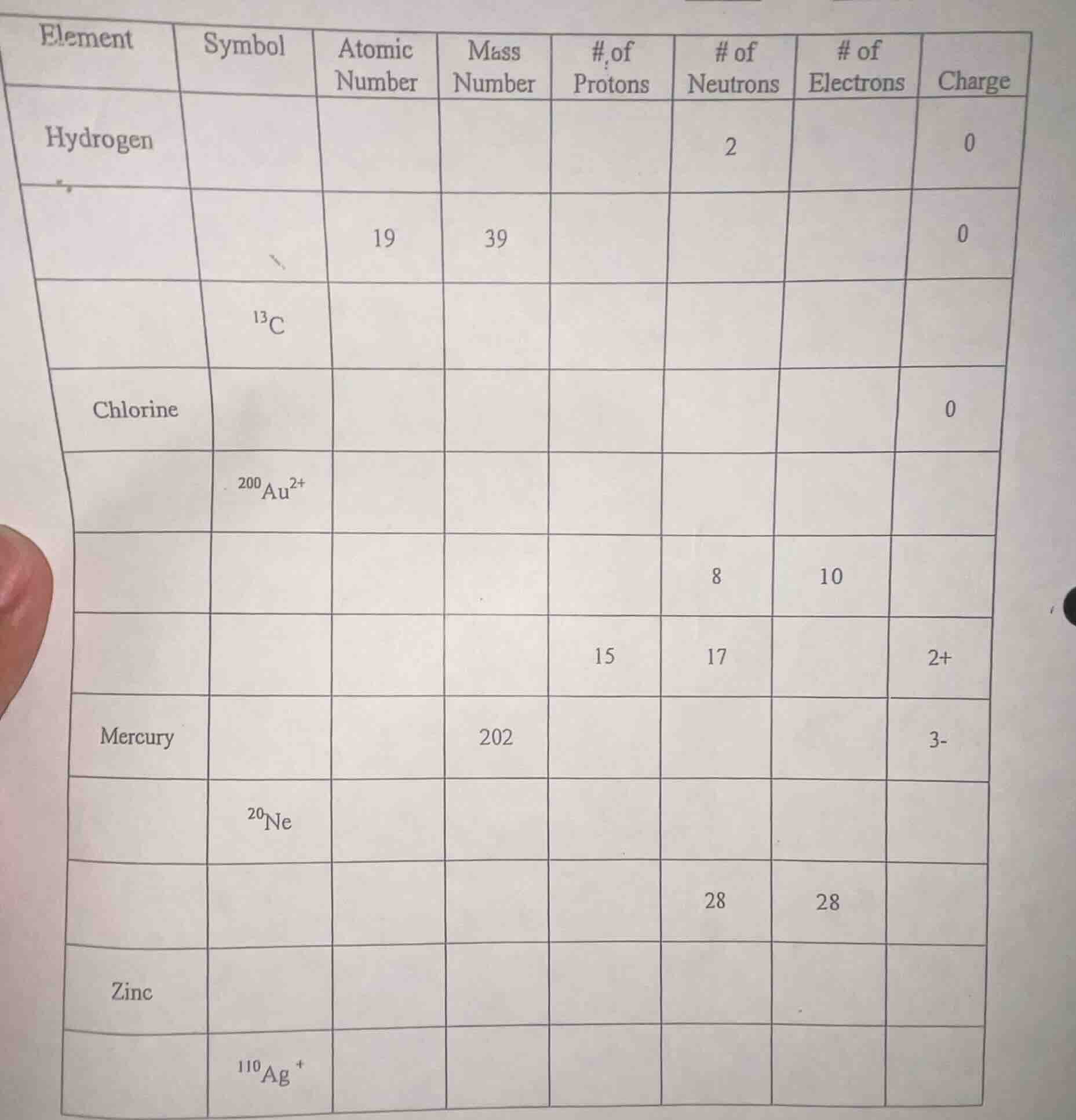
element | symbol | atomic number | mass number | # of protons | # of neutrons | # of electrons | charge
hydrogen | | | | | 2 | | 0
| 19 | 39 | 0 | ||||
| $^{13}$c |
chlorine | | | | | | | 0
| $^{200}$au$^{2+}$ | ||||||
| 8 | 10 | |||||
| 15 | 17 | 2+ |
mercury | | | 202 | | | | 3-
| $^{20}$ne | |||||
| 28 | 28 |
zinc | | | | | | |
| $^{110}$ag$^+$ |
Step1: Recall key atomic rules
- Atomic Number = # of Protons = # of Electrons (neutral atom)
- Mass Number = # of Protons + # of Neutrons
- Charge = # of Protons - # of Electrons
- Isotope notation: $^A_Z X^{c+}$, where $A$=Mass #, $Z$=Atomic #, $c$=charge
---
Row 1 (Hydrogen, charge=0, neutrons=2)
Step1: Find protons (neutral atom)
Charge=0, so #Protons=#Electrons. Hydrogen has atomic number 1, so #Protons=1
Step2: Calculate mass number
$\text{Mass Number} = 1 + 2 = 3$
Step3: Fill remaining values
Symbol: $^3\text{H}$, Atomic #=1, #Electrons=1
---
Row 2 (Atomic #=19, Mass #=39, charge=0)
Step1: Identify element & protons
Atomic #=19 = #Protons, element is Potassium (K)
Step2: Calculate neutrons
$\text{#Neutrons} = 39 - 19 = 20$
Step3: Find electrons (neutral)
#Electrons=#Protons=19
---
Row 3 ($^{13}\text{C}$)
Step1: Get atomic/mass numbers
Atomic # of C=6, Mass #=13, #Protons=6
Step2: Calculate neutrons
$\text{#Neutrons} = 13 - 6 = 7$
Step3: Find electrons (neutral)
Charge=0, so #Electrons=6, Element=Carbon
---
Row 4 (Chlorine, charge=0)
Step1: Get atomic number
Chlorine atomic #=17 = #Protons=#Electrons
Step2: Use most common mass #
Most common isotope is $^{35}\text{Cl}$, so Mass #=35
Step3: Calculate neutrons
$\text{#Neutrons} = 35 - 17 = 18$
Step4: Fill symbol
Symbol: $\text{Cl}$
---
Row 5 ($^{200}\text{Au}^{2+}$)
Step1: Get atomic/mass numbers
Au atomic #=79 = #Protons, Mass #=200
Step2: Calculate neutrons
$\text{#Neutrons} = 200 - 79 = 121$
Step3: Find electrons (2+ charge)
$\text{#Electrons} = 79 - 2 = 77$
Step4: Identify element & charge
Element=Gold, Charge=2+
---
Row 6 (#Neutrons=8, #Electrons=10)
Step1: Find charge
Assume neutral atomic #=8 (Oxygen), $\text{Charge} = 8 - 10 = 2-$
Step2: Calculate mass number
$\text{Mass Number} = 8 + 8 = 16$
Step3: Fill values
Element=Oxygen, Symbol: $^{16}\text{O}^{2-}$, Atomic #=8, #Protons=8
---
Row 7 (#Protons=15, #Neutrons=17, charge=2+)
Step1: Identify element
#Protons=15 = atomic #, element=Phosphorus (P)
Step2: Calculate mass number
$\text{Mass Number} = 15 + 17 = 32$
Step3: Find electrons
$\text{#Electrons} = 15 - 2 = 13$
Step4: Fill symbol
Symbol: $^{32}\text{P}^{2+}$, Atomic #=15
---
Row 8 (Mercury, Mass #=202, charge=3-)
Step1: Get atomic number
Hg atomic #=80 = #Protons
Step2: Calculate neutrons
$\text{#Neutrons} = 202 - 80 = 122$
Step3: Find electrons
$\text{#Electrons} = 80 + 3 = 83$
Step4: Fill symbol
Symbol: $^{202}\text{Hg}^{3-}$
---
Row 9 ($^{20}\text{Ne}$)
Step1: Get atomic number
Ne atomic #=10 = #Protons
Step2: Calculate neutrons
$\text{#Neutrons} = 20 - 10 = 10$
Step3: Find electrons (neutral)
#Electrons=10, Charge=0, Element=Neon
---
Row 10 (#Neutrons=28, #Electrons=28)
Step1: Assume neutral atom
#Protons=#Electrons=28, element=Nickel (Ni)
Step2: Calculate mass number
$\text{Mass Number} = 28 + 28 = 56$
Step3: Fill values
Atomic #=28, Charge=0, Symbol: $^{56}\text{Ni}$
---
Row 11 (Zinc)
Step1: Get atomic number
Zn atomic #=30 = #Protons
Step2: Use most common mass #
Most common isotope $^{65}\text{Zn}$, Mass #=65
Step3: Calculate neutrons
$\text{#Neutrons} = 65 - 30 = 35$
Step4: Fill values (neutral)
#Electrons=30, Charge=0, Symbol: $^{65}\text{Zn}$
---
Row 12 ($^{110}\text{Ag}^{+}$)
Step1: Get atomic number
Ag atomic #=47 = #Protons
Step2: Calculate neutrons
$\text{#Neutrons} = 110 - 47 = 63$
Step3: Find electrons (1+ charge)
$\text{#Electrons} = 47 - 1 = 46$
Step4: Fill values
Element…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Element | Symbol | Atomic Number | Mass Number | # of Protons | # of Neutrons | # of Electrons | Charge |
|---|---|---|---|---|---|---|---|
| Potassium | $\text{K}$ | 19 | 39 | 19 | 20 | 19 | 0 |
| Carbon | $^{13}\text{C}$ | 6 | 13 | 6 | 7 | 6 | 0 |
| Chlorine | $\text{Cl}$ | 17 | 35 | 17 | 18 | 17 | 0 |
| Gold | $^{200}\text{Au}^{2+}$ | 79 | 200 | 79 | 121 | 77 | 2+ |
| Oxygen | $^{16}\text{O}^{2-}$ | 8 | 16 | 8 | 8 | 10 | 2- |
| Phosphorus | $^{32}\text{P}^{2+}$ | 15 | 32 | 15 | 17 | 13 | 2+ |
| Mercury | $^{202}\text{Hg}^{3-}$ | 80 | 202 | 80 | 122 | 83 | 3- |
| Neon | $^{20}\text{Ne}$ | 10 | 20 | 10 | 10 | 10 | 0 |
| Nickel | $^{56}\text{Ni}$ | 28 | 56 | 28 | 28 | 28 | 0 |
| Zinc | $^{65}\text{Zn}$ | 30 | 65 | 30 | 35 | 30 | 0 |
| Silver | $^{110}\text{Ag}^{+}$ | 47 | 110 | 47 | 63 | 46 | 1+ |