QUESTION IMAGE
Question
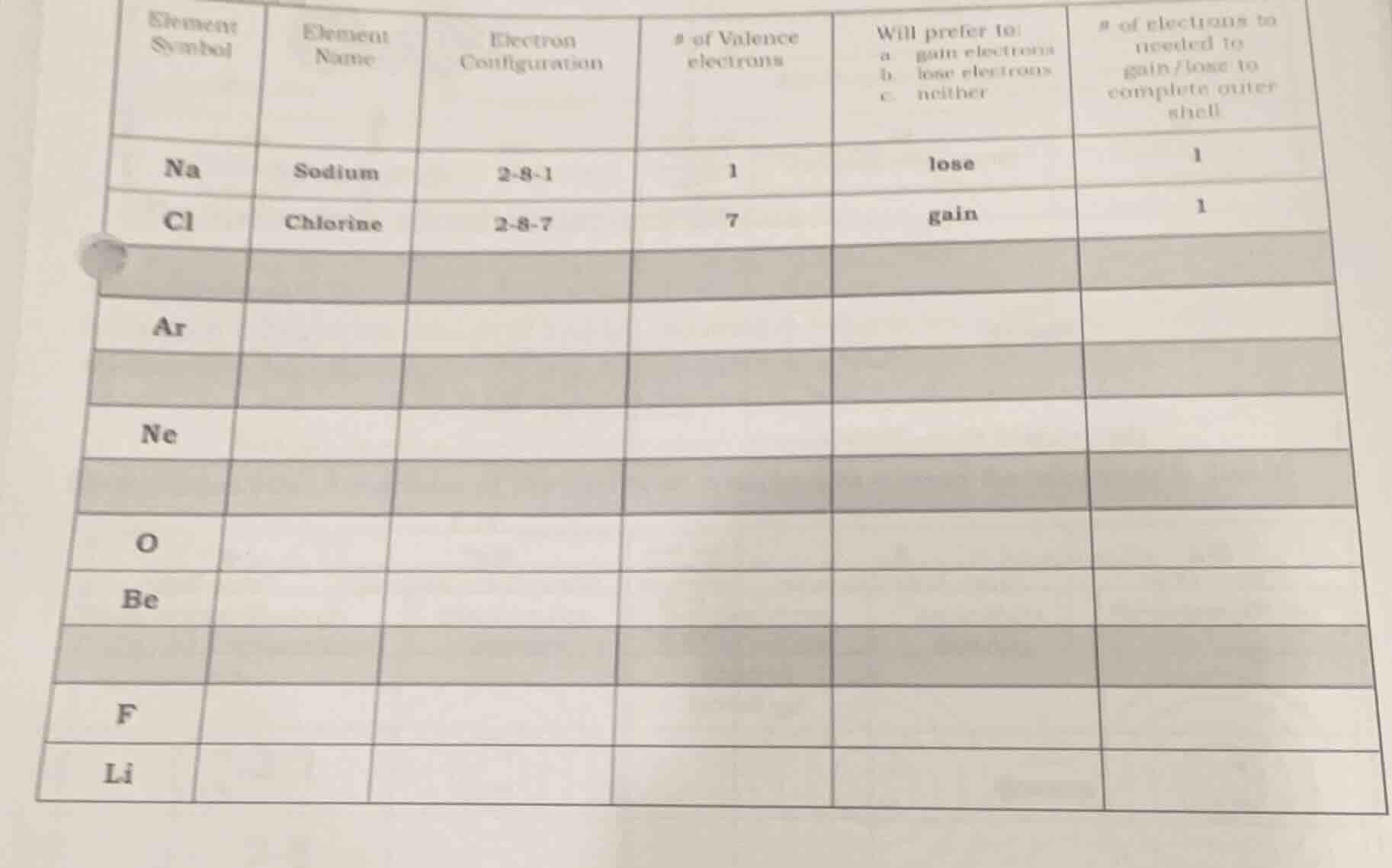
element symbol | element name | electron configuration | # of valence electrons | will prefer to: a. gain electrons b. lose electrons c. neither | # of electrons to needed to gain/lose to complete outer shell
na | sodium | 2 - 8 - 1 | 1 | lose | 1
cl | chlorine | 2 - 8 - 7 | 7 | gain | 1
ar | | | | |
ne | | | | |
o | | | | |
be | | | | |
f | | | | |
li | | | | |
Step1: Fill Arsenic (Ar) details
Element Name: Argon; Electron Configuration: 2-8-8; # of Valence electrons: 8; Prefer: neither; Electrons needed: 0
Step2: Fill Neon (Ne) details
Element Name: Neon; Electron Configuration: 2-8; # of Valence electrons: 8; Prefer: neither; Electrons needed: 0
Step3: Fill Oxygen (O) details
Element Name: Oxygen; Electron Configuration: 2-6; # of Valence electrons: 6; Prefer: gain; Electrons needed: 2
Step4: Fill Beryllium (Be) details
Element Name: Beryllium; Electron Configuration: 2-2; # of Valence electrons: 2; Prefer: lose; Electrons needed: 2
Step5: Fill Fluorine (F) details
Element Name: Fluorine; Electron Configuration: 2-7; # of Valence electrons: 7; Prefer: gain; Electrons needed: 1
Step6: Fill Lithium (Li) details
Element Name: Lithium; Electron Configuration: 2-1; # of Valence electrons: 1; Prefer: lose; Electrons needed: 1
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Element Symbol | Element Name | Electron Configuration | # of Valence electrons | Will prefer to: a. gain electrons b. lose electrons c. neither | # of electrons to needed to gain/lose to complete outer shell |
|---|---|---|---|---|---|
| Cl | Chlorine | 2-8-7 | 7 | gain | 1 |
| Ar | Argon | 2-8-8 | 8 | neither | 0 |
| Ne | Neon | 2-8 | 8 | neither | 0 |
| O | Oxygen | 2-6 | 6 | gain | 2 |
| Be | Beryllium | 2-2 | 2 | lose | 2 |
| F | Fluorine | 2-7 | 7 | gain | 1 |
| Li | Lithium | 2-1 | 1 | lose | 1 |