QUESTION IMAGE
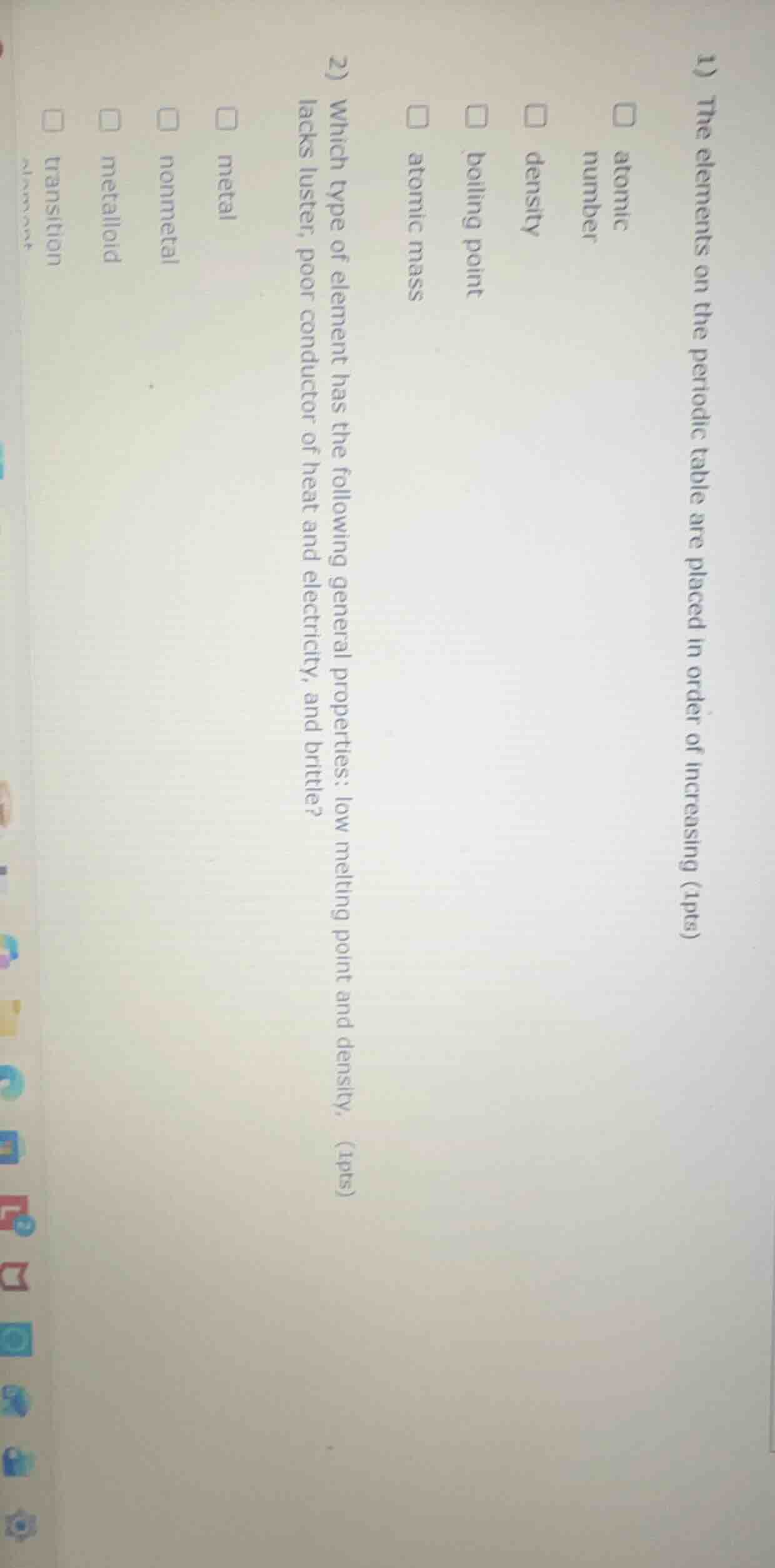
Question
- the elements on the periodic table are placed in order of increasing (1pts)
□ atomic number
□ density
□ boiling point
□ atomic mass
- which type of element has the following general properties: low melting point and density, lacks luster, poor conductor of heat and electricity, and brittle? (1pts)
□ metal
□ nonmetal
□ metalloid
□ transition element
Brief Explanations
- The modern periodic table organizes elements by the number of protons in their nucleus, which is the atomic number, in ascending order. Other properties like mass or density do not define the core ordering.
- Nonmetals typically have low melting points/density, are brittle, lack metallic luster, and are poor conductors of heat and electricity, matching the described properties. Metals are good conductors and lustrous; metalloids have intermediate properties; transition metals are a subset of metals.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- atomic number
- nonmetal