QUESTION IMAGE
Question
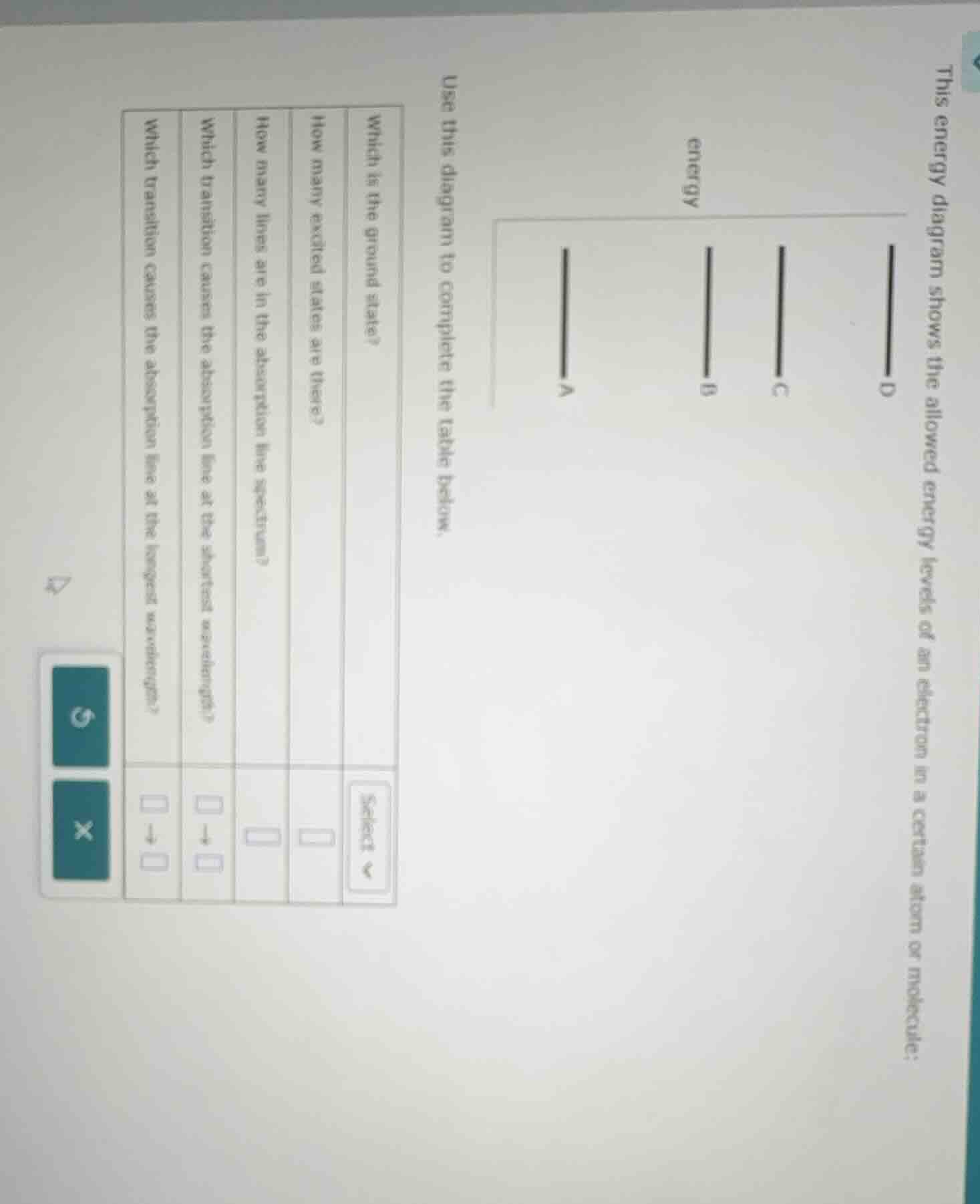
this energy diagram shows the allowed energy levels of an electron in a certain atom or molecule: use this diagram to complete the table below. which is the ground state? how many excited states are there? how many lines are in the absorption line spectrum? which transition causes the absorption line at the shortest wavelength? which transition causes the absorption line at the longest wavelength?
1. Which is the ground state?
The ground state is the lowest - energy state. In the energy diagram, the level with the lowest energy (the bottom - most level) is the ground state. Among the levels D, C, B, A, D has the lowest energy (since energy increases as we go up the diagram), so D is the ground state.
Excited states are energy levels above the ground state. The ground state is D. The levels above D are C, B, and A. So we count the number of levels above the ground state. There are 3 levels (C, B, A) above D, so there are 3 excited states.
Absorption lines occur when an electron transitions from the ground state (D) to an excited state. The possible transitions from D are to C, to B, and to A. Each transition corresponds to one absorption line. So the number of absorption lines is equal to the number of possible transitions from the ground state to excited states. Since there are 3 excited states (C, B, A) accessible from D, there are 3 absorption lines.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D