QUESTION IMAGE
Question
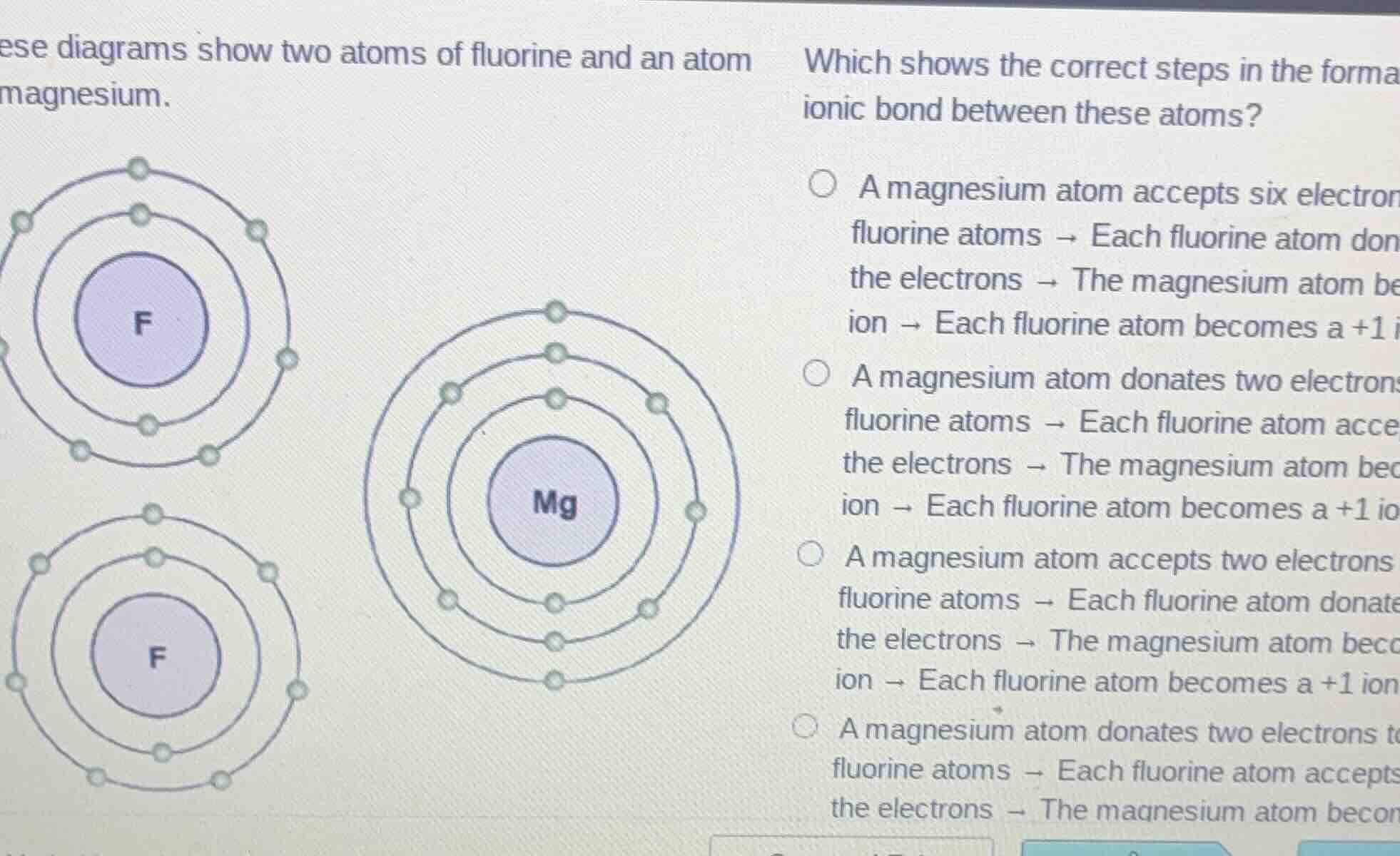
ese diagrams show two atoms of fluorine and an atom magnesium. which shows the correct steps in the forma ionic bond between these atoms? a magnesium atom accepts six electron fluorine atoms → each fluorine atom don the electrons → the magnesium atom be ion → each fluorine atom becomes a +1 i a magnesium atom donates two electron fluorine atoms → each fluorine atom acce the electrons → the magnesium atom bec ion → each fluorine atom becomes a +1 io a magnesium atom accepts two electrons fluorine atoms → each fluorine atom donate the electrons → the magnesium atom bec ion → each fluorine atom becomes a +1 ion a magnesium atom donates two electrons t fluorine atoms → each fluorine atom accepts the electrons → the magnesium atom becon
- Recall the electron configuration and ionic bonding rules: Magnesium (Mg) has 2 valence electrons and tends to lose them (donate) to achieve a stable octet. Fluorine (F) has 7 valence electrons and tends to gain 1 electron (accept) to achieve a stable octet.
- Analyze each option:
- Option 1: Magnesium accepting electrons is wrong (Mg donates, not accepts). Also, fluorine donating electrons is wrong (F accepts). Eliminate.
- Option 2: Magnesium donates 2 electrons (one to each F atom, since there are two F atoms). Each F accepts 1 electron. After donation, Mg loses 2 electrons, becoming \( \text{Mg}^{2+} \) (a +2 ion), and each F gains 1 electron, becoming \( \text{F}^- \) (a -1 ion, but the option mentions +1 which is incorrect? Wait, no—wait, the option's last part might have a typo, but the first steps: Mg donates 2 (one to each F), each F accepts 1. This matches the electron transfer logic (Mg is a metal, donates; F is a non - metal, accepts).
- Option 3: Magnesium accepting electrons and fluorine donating is wrong (opposite of their electronegativity - based behavior). Eliminate.
- Option 4: The last part is cut off, but from the first two steps, Option 2 has the correct electron donation (Mg donates) and acceptance (F accepts) in terms of the direction (even if the ion charge description in the option has a possible typo, the electron transfer direction is correct compared to others).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The correct option is the one where "A magnesium atom donates two electrons to fluorine atoms → Each fluorine atom accepts the electrons → The magnesium atom becomes a +2 ion → Each fluorine atom becomes a - 1 ion" (assuming the option's last part about +1 was a typo, and this is the second option in the list). So the answer is the second option (the one starting with "A magnesium atom donates two electrons...").