QUESTION IMAGE
Question
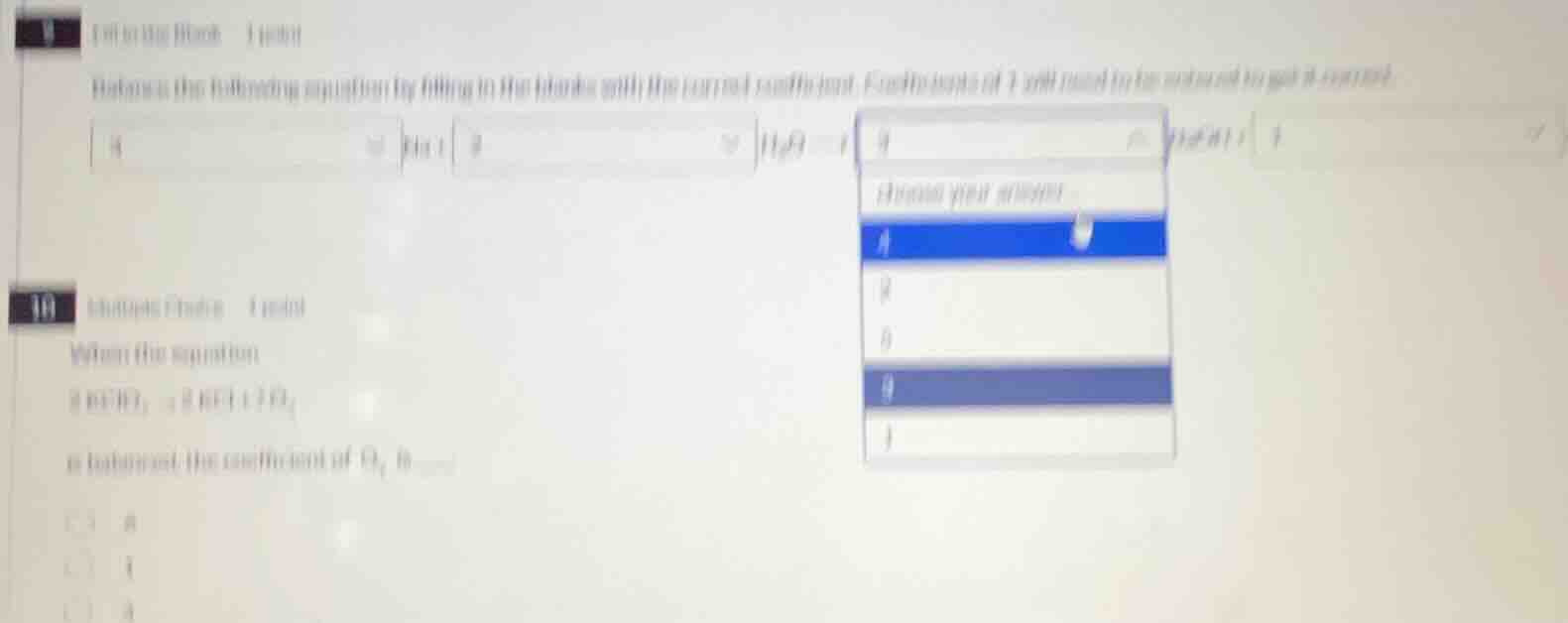
9 fill in the blank 1 point
balance the following equation by filling in the blanks with the correct coefficient. coefficients of 1 will need to be entered to get it correct.
k + h₂o = koh + h₂
choose your answer:
4
2
6
9
1
10 multiple choice 1 point
when the equation
8 kclo₃ → 8 kcl + o₂
is balanced, the coefficient of o₂ is_____
a. 8
b. 1
c. 4
Step1: Balance Cl atoms
Count Cl on right: $2\text{KCl} + 3\text{Cl}_2$ gives $2 + 6 = 8$ Cl. So left $\text{KClO}_3$ needs coefficient 4: $4\text{KClO}_3
ightarrow \text{KCl} + \text{Cl}_2 + \text{O}_2$
Step2: Balance K atoms
Left has 4 K, so right $\text{KCl}$ gets coefficient 4: $4\text{KClO}_3
ightarrow 4\text{KCl} + \text{Cl}_2 + \text{O}_2$
Step3: Balance Cl (remaining)
Left Cl: 4, right KCl has 4, so $\text{Cl}_2$ coefficient is $\frac{4-4}{2}=0$? Correct: $4\text{KClO}_3
ightarrow 4\text{KCl} + 6\text{O}_2$ (now O: left $4\times3=12$, right $6\times2=12$)
Step4: Find $\text{O}_2$ coefficient
From balanced equation, $\text{O}_2$ coefficient is 6.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
6