QUESTION IMAGE
Question
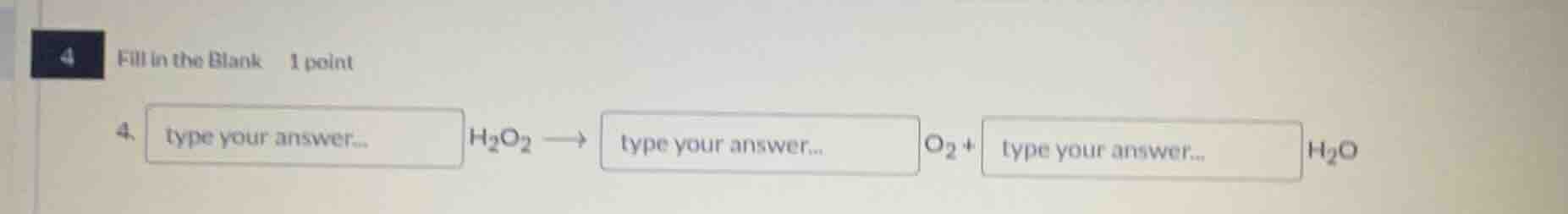
4 fill in the blank 1 point 4. type your answer... h₂o₂ → type your answer... o₂ + type your answer... h₂o
Step1: Balance the hydrogen peroxide decomposition reaction. The reaction is the decomposition of hydrogen peroxide (\(H_2O_2\)) into oxygen (\(O_2\)) and water (\(H_2O\)). First, write the unbalanced equation: \(H_2O_2
ightarrow O_2 + H_2O\).
Step2: Balance the oxygen and hydrogen atoms. Let's start with oxygen. On the left, we have 2 O in \(H_2O_2\). On the right, in \(O_2\) we have 2 O and in \(H_2O\) we have 1 O per molecule. Let's balance the equation by putting coefficients. The balanced equation is \(2H_2O_2
ightarrow O_2 + 2H_2O\). So the coefficients are 2, 1, and 2 (but the middle one for \(O_2\) is 1, but usually we write the coefficients. Wait, the blanks: first blank is coefficient for \(H_2O_2\), second for \(O_2\), third for \(H_2O\). Wait, the reaction is decomposition of \(H_2O_2\), so the balanced equation is \(2H_2O_2
ightarrow 1O_2 + 2H_2O\). So the coefficients are 2, 1, 2. But maybe the question is about the reaction type? No, fill in the blanks with coefficients. Wait, the first blank: coefficient for \(H_2O_2\), second for \(O_2\), third for \(H_2O\). So:
First blank: 2 (coefficient of \(H_2O_2\))
Second blank: 1 (coefficient of \(O_2\))
Third blank: 2 (coefficient of \(H_2O\))
Wait, but let's check the atoms. For \(H\): left side 22=4 H, right side 22=4 H (in \(H_2O\)). For O: left side 22=4 O, right side 12 + 2*1=4 O. So balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
First blank: 2, Second blank: 1, Third blank: 2