QUESTION IMAGE
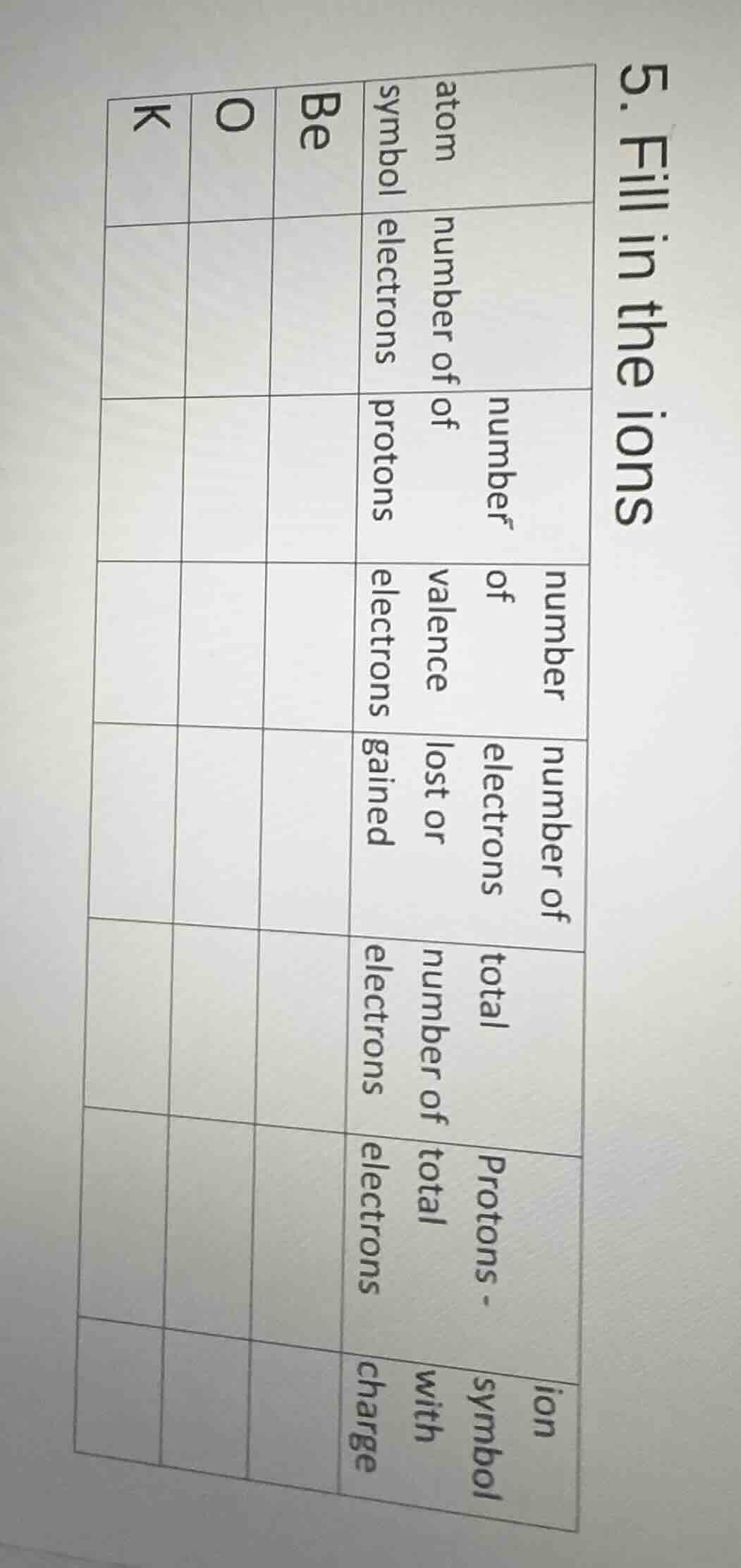
Question
- fill in the ions
| atom symbol | number of electrons | number of protons | number of valence electrons | number of electrons lost or gained | total number of electrons | protons - total electrons | ion symbol with charge | |||
| be | ||||||||||
| o | ||||||||||
| k |
To solve this, we use the following rules:
- Atomic number (protons) = number of protons in neutral atom.
- In neutral atom, electrons = protons. For ions:
- Cations (lose electrons): electrons = protons - lost electrons.
- Anions (gain electrons): electrons = protons + gained electrons.
- Charge = protons - electrons.
For Be (Beryllium)
Step 1: Protons (Atomic Number)
Be has atomic number 4, so protons = 4.
Step 2: Neutral Electrons
Neutral Be: electrons = protons = 4.
Step 3: Valence Electrons
Be is in Group 2, so valence electrons = 2.
Step 4: Electrons Lost/Gained (Ion)
Be forms \( \text{Be}^{2+} \) (loses 2 electrons). So electrons lost = 2.
Step 5: Total Electrons in Ion
Electrons = protons - lost = \( 4 - 2 = 2 \).
Step 6: Protons - Electrons (Charge)
Charge = \( 4 - 2 = +2 \).
Step 7: Ion Symbol
\( \text{Be}^{2+} \).
For O (Oxygen)
Step 1: Protons (Atomic Number)
O has atomic number 8, so protons = 8.
Step 2: Neutral Electrons
Neutral O: electrons = protons = 8.
Step 3: Valence Electrons
O is in Group 16, so valence electrons = 6 (or 2, but typically 6 for non - metal; gains 2 to fill octet).
Step 4: Electrons Lost/Gained (Ion)
O forms \( \text{O}^{2-} \) (gains 2 electrons). So electrons gained = 2.
Step 5: Total Electrons in Ion
Electrons = protons + gained = \( 8 + 2 = 10 \).
Step 6: Protons - Electrons (Charge)
Charge = \( 8 - 10 = -2 \).
Step 7: Ion Symbol
\( \text{O}^{2-} \).
For K (Potassium)
Step 1: Protons (Atomic Number)
K has atomic number 19, so protons = 19.
Step 2: Neutral Electrons
Neutral K: electrons = protons = 19.
Step 3: Valence Electrons
K is in Group 1, so valence electrons = 1.
Step 4: Electrons Lost/Gained (Ion)
K forms \( \text{K}^+ \) (loses 1 electron). So electrons lost = 1.
Step 5: Total Electrons in Ion
Electrons = protons - lost = \( 19 - 1 = 18 \).
Step 6: Protons - Electrons (Charge)
Charge = \( 19 - 18 = +1 \).
Step 7: Ion Symbol
\( \text{K}^+ \).
Filled Table (Summary)
| Atom Symbol | Number of Electrons (Neutral) | Number of Protons | Number of Valence Electrons | Number of Electrons Lost/Gained | Total Electrons (Ion) | Protons - Electrons (Charge) | Ion Symbol with Charge |
|---|---|---|---|---|---|---|---|
| O | 8 | 8 | 6 | Gained 2 | 10 | -2 | \( \text{O}^{2-} \) |
| K | 19 | 19 | 1 | Lost 1 | 18 | +1 | \( \text{K}^+ \) |
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The filled table (key values) is as above. For each ion:
- Be: \( \text{Be}^{2+} \) (protons = 4, electrons in ion = 2, charge = +2)
- O: \( \text{O}^{2-} \) (protons = 8, electrons in ion = 10, charge = -2)
- K: \( \text{K}^+ \) (protons = 19, electrons in ion = 18, charge = +1)