QUESTION IMAGE
Question
forensic analysis
a forensic chemist intern is trying to determine
information about a sample of a dark reddish-purple
hair dye found at a crime scene to identify the brand.
one of the tests involves a spectrophotometric
analysis of the dye color and concentration.
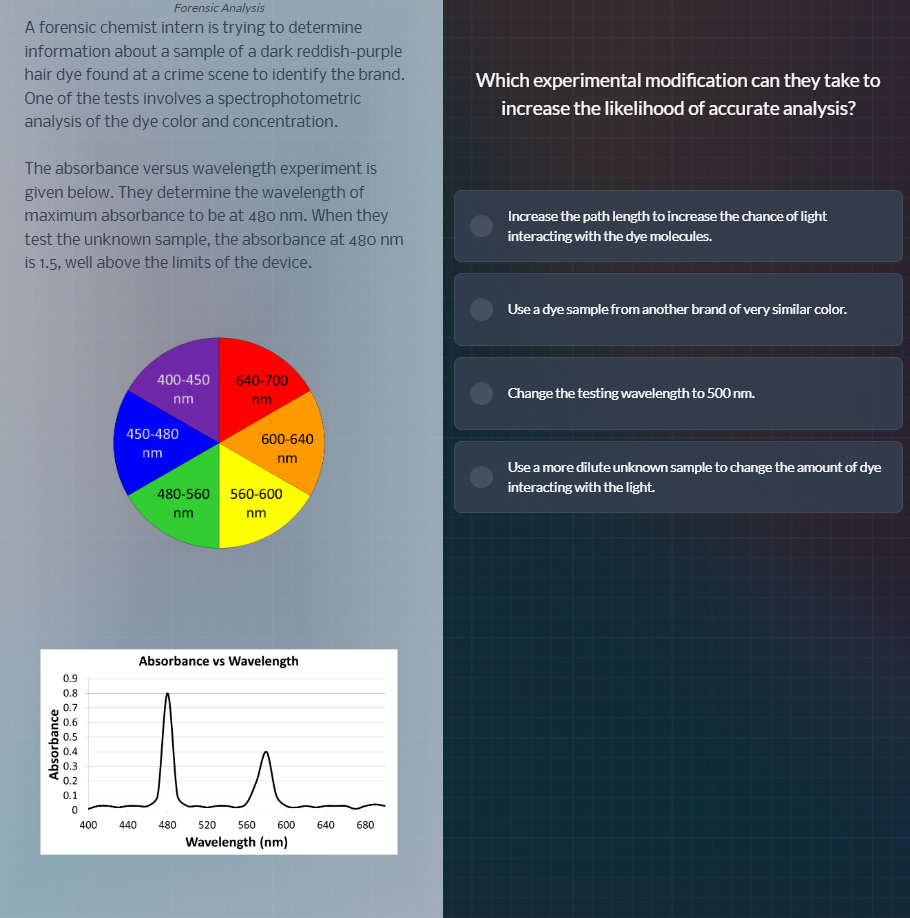
the absorbance versus wavelength experiment is
given below. they determine the wavelength of
maximum absorbance to be at 480 nm. when they
test the unknown sample, the absorbance at 480 nm
is 1.5, well above the limits of the device.
which experimental modification can they take to
increase the likelihood of accurate analysis?
increase the path length to increase the chance of light
interacting with the dye molecules.
use a dye sample from another brand of very similar color.
change the testing wavelength to 500 nm.
use a more dilute unknown sample to change the amount of dye
interacting with the light.
The issue is that the absorbance (1.5) is beyond the device's limits, making readings inaccurate. Beer-Lambert Law ($A = \epsilon l c$) shows absorbance depends on molar absorptivity ($\epsilon$), path length ($l$), and concentration ($c$). Increasing path length or using a non-optimal wavelength would raise or keep absorbance too high. Using a different brand is irrelevant to fixing the over-limit reading. Diluting the sample lowers concentration ($c$), which reduces absorbance to a measurable, accurate range for the device.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Use a more dilute unknown sample to change the amount of dye interacting with the light.