QUESTION IMAGE
Question
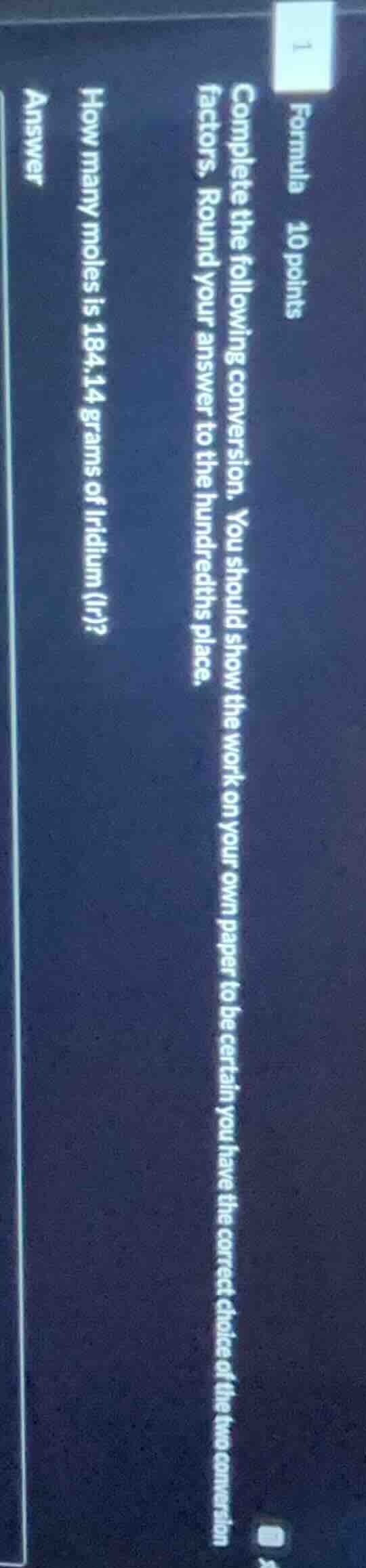
formula 10 points complete the following conversion. you should show the work on your own paper to be certain you have the correct choice of the two conversion factors. round your answer to the hundredths place. how many moles is 184.14 grams of iridium (ir)? answer
Step 1: Recall the molar mass of Iridium (Ir)
The molar mass of iridium (Ir) is approximately \( 192.22 \, \text{g/mol} \). This value is obtained from the periodic table, where the atomic mass of iridium is listed, and for a single atom, the atomic mass in atomic mass units (amu) is numerically equal to the molar mass in grams per mole (g/mol) for one mole of atoms.
Step 2: Use the formula for moles
The formula to calculate the number of moles (\( n \)) is given by:
\[
n = \frac{\text{mass (in grams)}}{\text{molar mass (in g/mol)}}
\]
We are given the mass of iridium as \( 184.14 \, \text{g} \) and the molar mass as \( 192.22 \, \text{g/mol} \). Substituting these values into the formula:
\[
n = \frac{184.14}{192.22}
\]
Step 3: Perform the calculation
Now, we calculate the value of the above expression:
\[
\frac{184.14}{192.22} \approx 0.958
\]
Step 4: Round to the hundredths place
Rounding \( 0.958 \) to the hundredths place (two decimal places), we look at the third decimal digit, which is \( 8 \). Since \( 8 \geq 5 \), we round up the second decimal digit. So, \( 0.958 \approx 0.96 \) (when rounded to the hundredths place).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The number of moles of iridium in \( 184.14 \) grams is approximately \( \boldsymbol{0.96} \) moles.