QUESTION IMAGE
Question
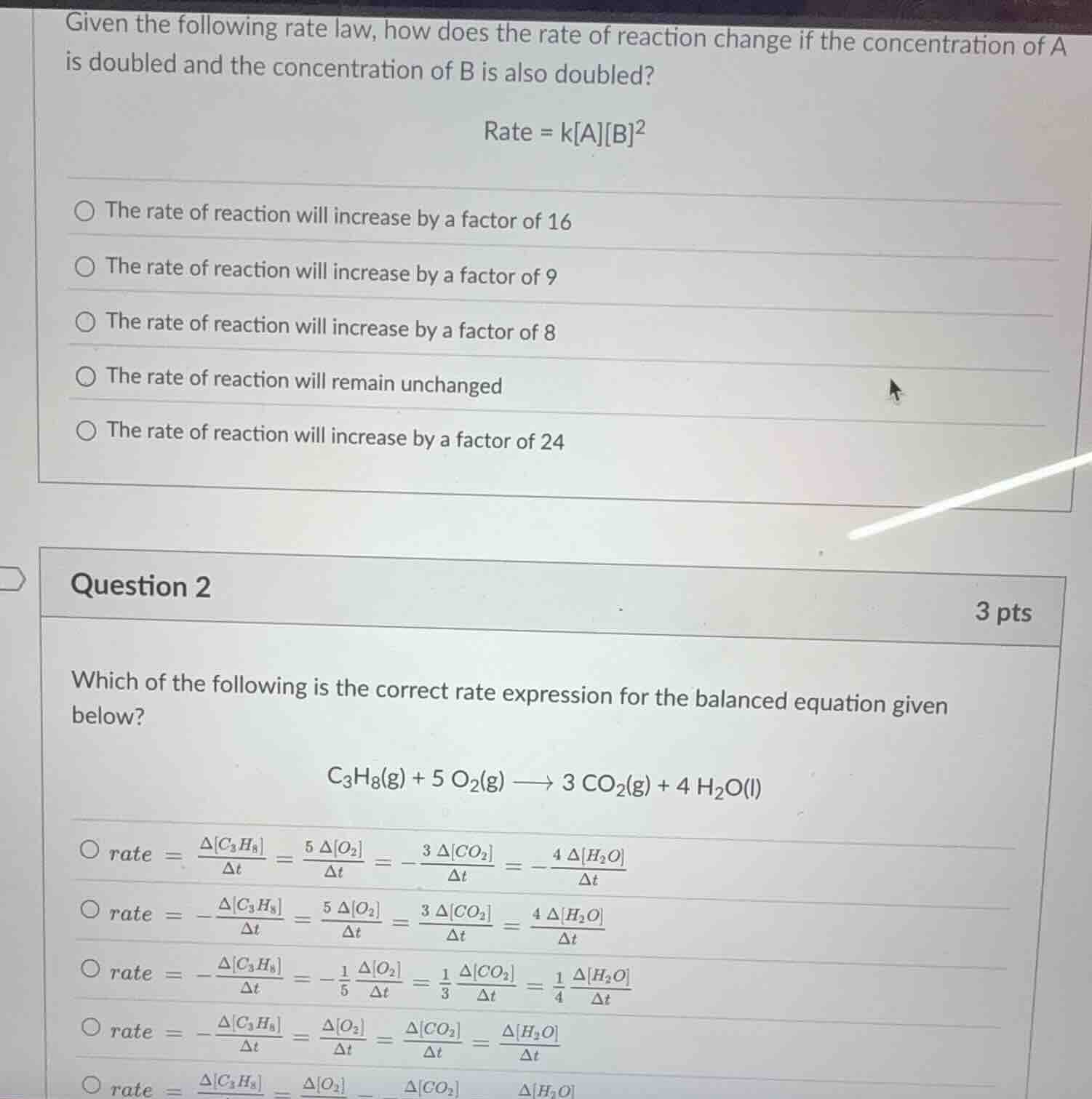
given the following rate law, how does the rate of reaction change if the concentration of a is doubled and the concentration of b is also doubled? rate = kab² the rate of reaction will increase by a factor of 16 the rate of reaction will increase by a factor of 9 the rate of reaction will increase by a factor of 8 the rate of reaction will remain unchanged the rate of reaction will increase by a factor of 24 question 2 3 pts which of the following is the correct rate expression for the balanced equation given below? c₃h₈(g) + 5 o₂(g) ⟶ 3 co₂(g) + 4 h₂o(l) rate = (δc₃h₈)/δt = (5 δo₂)/δt = - (3 δco₂)/δt = - (4 δh₂o)/δt rate = - (δc₃h₈)/δt = (5 δo₂)/δt = (3 δco₂)/δt = (4 δh₂o)/δt rate = - (δc₃h₈)/δt = - (1/5) (δo₂)/δt = (1/3) (δco₂)/δt = (1/4) (δh₂o)/δt rate = - (δc₃h₈)/δt = (δo₂)/δt = (δco₂)/δt = (δh₂o)/δt rate = (δc₃h₈)/δt = (δo₂)/δt - (δco₂)/δt (δh₂o)/δt
Question 1
Step1: Define initial rate
Let initial concentration of \( A \) be \([A]_1\) and \( B \) be \([B]_1\). Initial rate \( r_1 = k[A]_1[B]_1^2 \).
Step2: Define new concentrations
After doubling, \([A]_2 = 2[A]_1\) and \([B]_2 = 2[B]_1\).
Step3: Calculate new rate
New rate \( r_2 = k[A]_2[B]_2^2 = k(2[A]_1)(2[B]_1)^2 \).
Simplify: \( r_2 = k \times 2[A]_1 \times 4[B]_1^2 = 8k[A]_1[B]_1^2 \).
Since \( r_1 = k[A]_1[B]_1^2 \), \( r_2 = 8r_1 \).
For a reaction \( aA + bB
ightarrow cC + dD \), the rate expression is \( \text{rate} = -\frac{1}{a}\frac{\Delta [A]}{\Delta t}=-\frac{1}{b}\frac{\Delta [B]}{\Delta t}=\frac{1}{c}\frac{\Delta [C]}{\Delta t}=\frac{1}{d}\frac{\Delta [D]}{\Delta t} \). For \( \ce{C3H8(g) + 5 O2(g) -> 3 CO2(g) + 4 H2O(l)} \), \( a = 1, b = 5, c = 3, d = 4 \). Reactants have negative signs (concentration decreases), products positive. So \( \text{rate} = -\frac{\Delta [\ce{C3H8}]}{\Delta t}=-\frac{1}{5}\frac{\Delta [\ce{O2}]}{\Delta t}=\frac{1}{3}\frac{\Delta [\ce{CO2}]}{\Delta t}=\frac{1}{4}\frac{\Delta [\ce{H2O}]}{\Delta t} \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The rate of reaction will increase by a factor of 8