QUESTION IMAGE
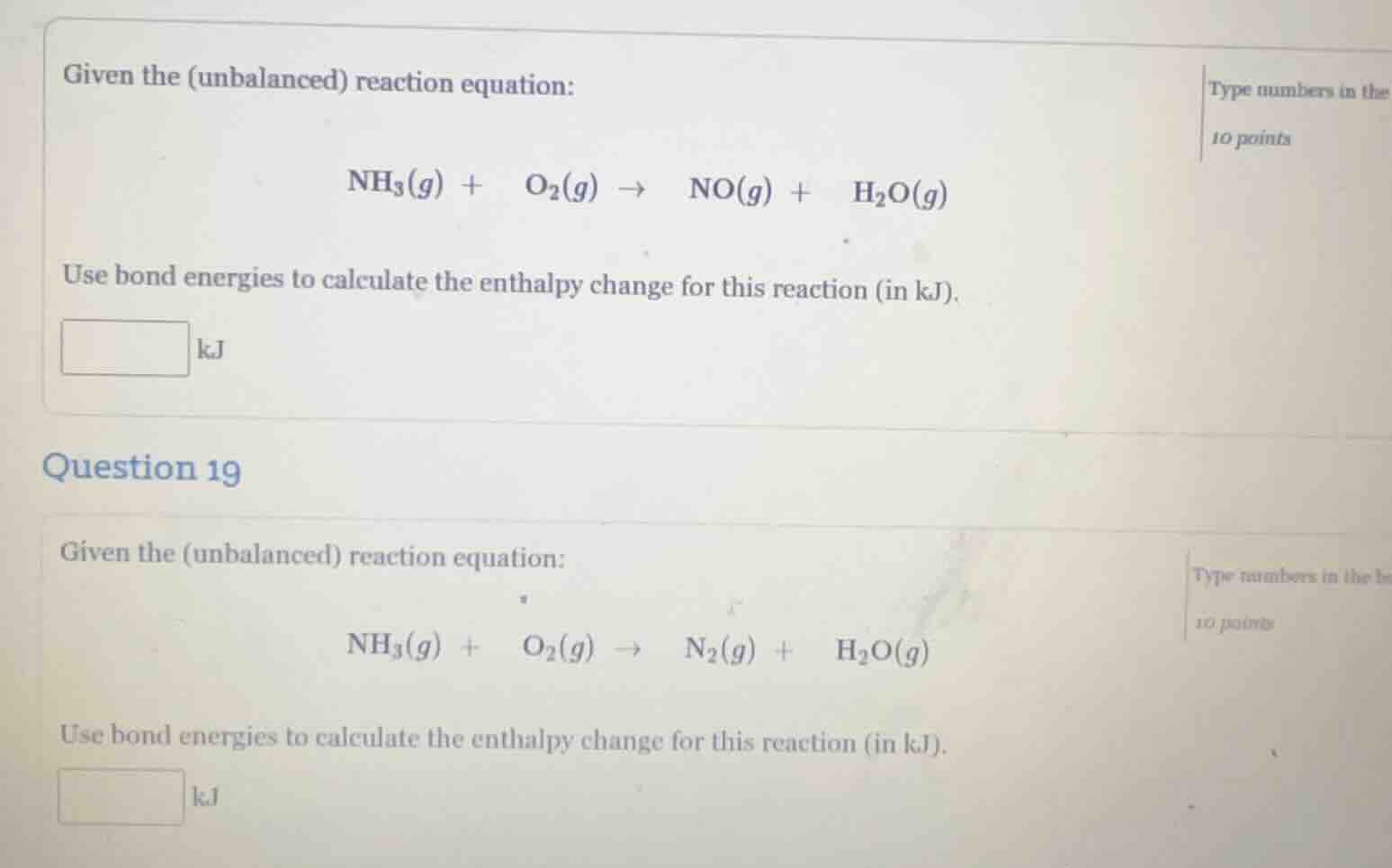
Question
given the (unbalanced) reaction equation: nh₃(g) + o₂(g) → no(g) + h₂o(g) use bond energies to calculate the enthalpy change for this reaction (in kj). kj question 19 given the (unbalanced) reaction equation: nh₃(g) + o₂(g) → n₂(g) + h₂o(g) use bond energies to calculate the enthalpy change for this reaction (in kj). kj
Common Bond Energies (used for calculations):
$E(\text{N-H}) = 391$ kJ/mol, $E(\text{O=O}) = 498$ kJ/mol, $E(\text{N=O}) = 607$ kJ/mol, $E(\text{O-H}) = 464$ kJ/mol, $E(\text{N≡N}) = 945$ kJ/mol
---
For Reaction 1: $\text{NH}_3(g) + \text{O}_2(g)
ightarrow \text{NO}(g) + \text{H}_2\text{O}(g)$
Step1: Balance the reaction
$4\text{NH}_3(g) + 5\text{O}_2(g)
ightarrow 4\text{NO}(g) + 6\text{H}_2\text{O}(g)$
Step2: Calculate bond breakage energy
Break 12 N-H bonds + 5 O=O bonds:
$\Delta H_{\text{break}} = (12 \times 391) + (5 \times 498) = 4692 + 2490 = 7182$ kJ
Step3: Calculate bond formation energy
Form 4 N=O bonds + 12 O-H bonds (energy released, negative):
$\Delta H_{\text{form}} = -(4 \times 607) - (12 \times 464) = -2428 - 5568 = -7990$ kJ
Step4: Compute total enthalpy change
$\Delta H_{\text{rxn}} = \Delta H_{\text{break}} + \Delta H_{\text{form}} = 7182 - 7990 = -902$ kJ
---
For Reaction 2: $\text{NH}_3(g) + \text{O}_2(g)
ightarrow \text{N}_2(g) + \text{H}_2\text{O}(g)$
Step1: Balance the reaction
$4\text{NH}_3(g) + 3\text{O}_2(g)
ightarrow 2\text{N}_2(g) + 6\text{H}_2\text{O}(g)$
Step2: Calculate bond breakage energy
Break 12 N-H bonds + 3 O=O bonds:
$\Delta H_{\text{break}} = (12 \times 391) + (3 \times 498) = 4692 + 1494 = 6186$ kJ
Step3: Calculate bond formation energy
Form 2 N≡N bonds + 12 O-H bonds (energy released, negative):
$\Delta H_{\text{form}} = -(2 \times 945) - (12 \times 464) = -1890 - 5568 = -7458$ kJ
Step4: Compute total enthalpy change
$\Delta H_{\text{rxn}} = \Delta H_{\text{break}} + \Delta H_{\text{form}} = 6186 - 7458 = -1266$ kJ
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- First reaction: $-902$ kJ
- Second reaction: $-1266$ kJ