QUESTION IMAGE
Question
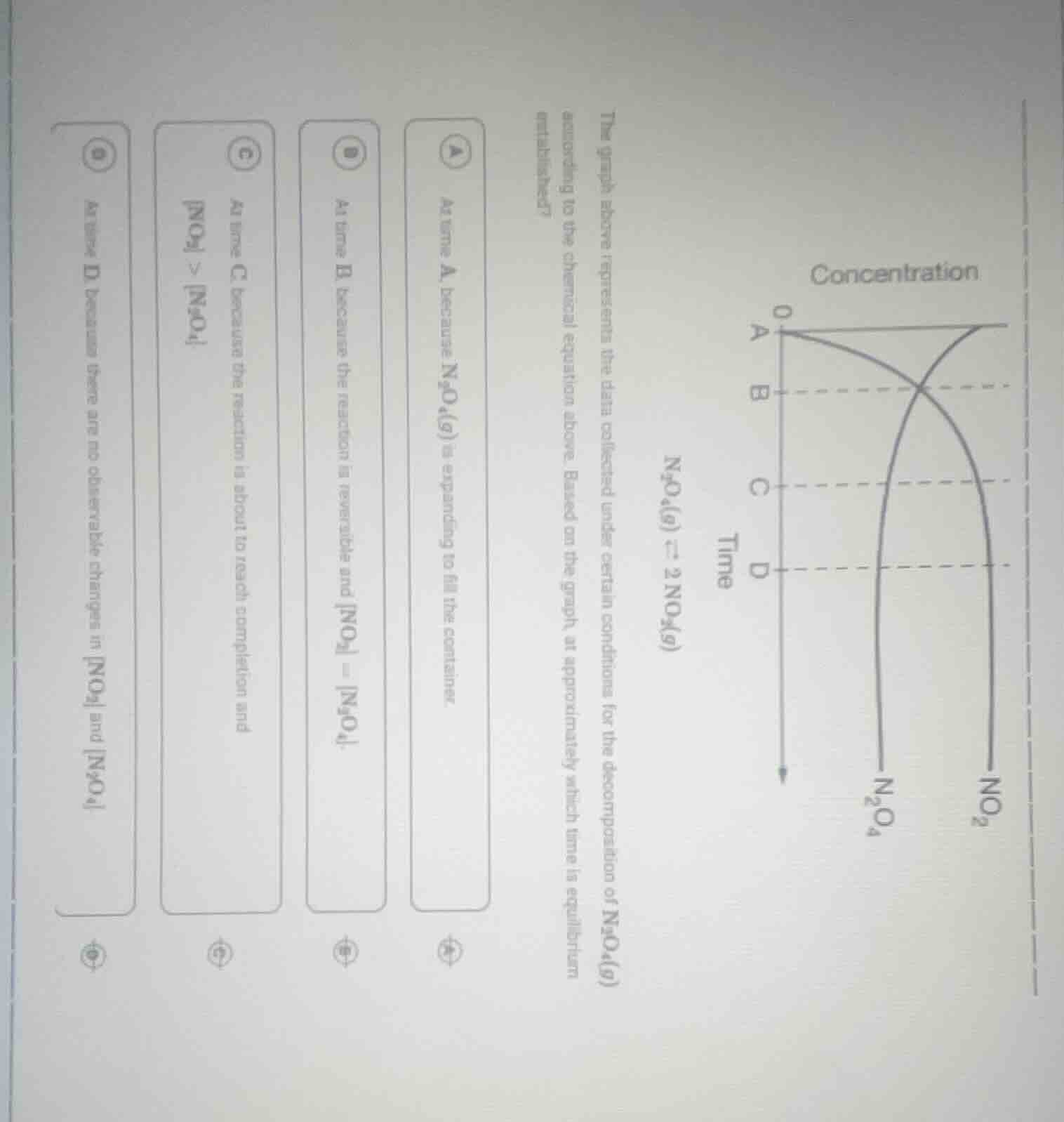
the graph above represents the data collected under certain conditions for the decomposition of n₂o₄(g) according to the chemical equation above. based on the graph, at approximately which time is equilibrium established? a at time a, because n₂o₄(g) is expanding to fill the container. b at time b, because the reaction is reversible and no₂ = n₂o₄. c at time c, because the reaction is about to reach completion and no₂ > n₂o₄. d at time d, because there are no observable changes in no₂ and n₂o₄.
To determine when equilibrium is established for the reaction $\ce{N_{2}O_{4}(g)
ightleftharpoons 2NO_{2}(g)}$, we analyze the concentration - time graph:
- Option A: At time A, the reaction has just started. $\ce{N_{2}O_{4}}$ is decomposing, but equilibrium is not reached yet because the concentrations of reactants and products are still changing rapidly. The idea of "expanding to fill the container" is not relevant to the concept of chemical equilibrium here. So, A is incorrect.
- Option B: At time B, the concentrations of $\ce{NO_{2}}$ and $\ce{N_{2}O_{4}}$ are equal. However, chemical equilibrium is not defined by equal concentrations of reactants and products. It is defined by the state where the rate of the forward reaction equals the rate of the reverse reaction, and the concentrations of reactants and products remain constant (not necessarily equal). So, B is incorrect.
- Option C: At time C, the reaction is still proceeding. The concentration of $\ce{NO_{2}}$ is greater than that of $\ce{N_{2}O_{4}}$, but the concentrations are still changing, which means the reaction has not reached equilibrium. Equilibrium requires that the concentrations stop changing. So, C is incorrect.
- Option D: At time D, the concentrations of $\ce{NO_{2}}$ and $\ce{N_{2}O_{4}}$ become constant (no observable changes in their concentrations). This is the characteristic of a chemical equilibrium, where the rate of the forward reaction ($\ce{N_{2}O_{4}}$ decomposing to $\ce{NO_{2}}$) equals the rate of the reverse reaction ($\ce{NO_{2}}$ combining to form $\ce{N_{2}O_{4}}$). So, D is correct.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. At time D, because there are no observable changes in $\boldsymbol{[\ce{NO_{2}}]}$ and $\boldsymbol{[\ce{N_{2}O_{4}}]}$