QUESTION IMAGE
Question
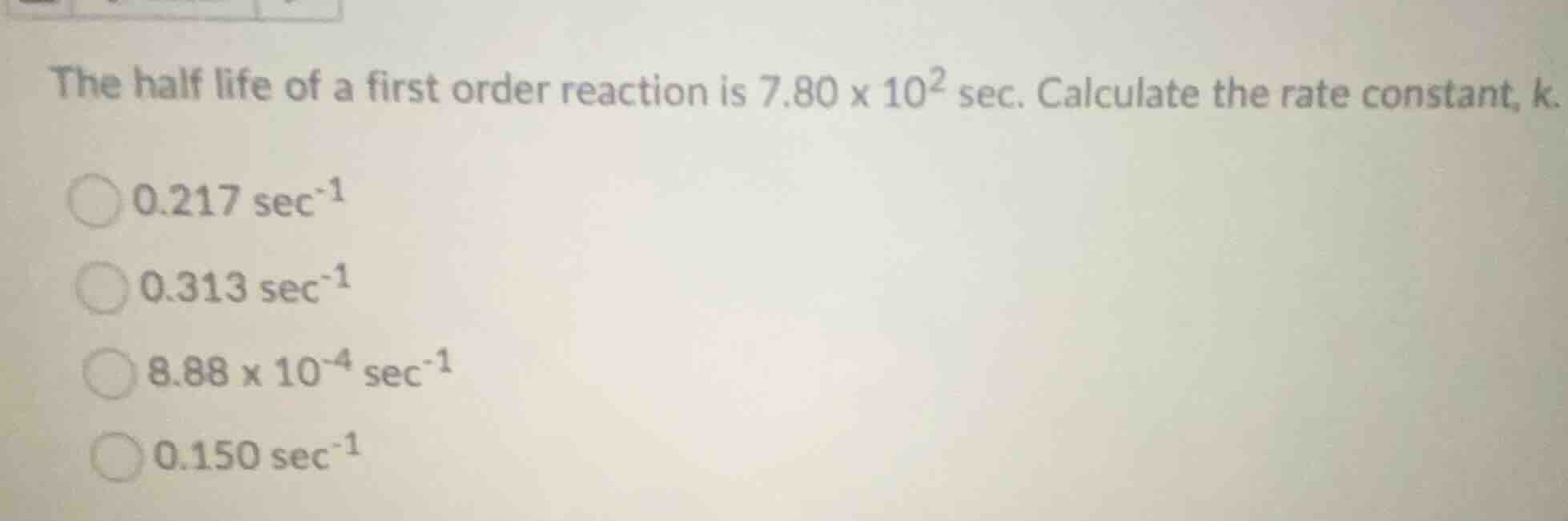
the half life of a first order reaction is $7.80 \times 10^{2}$ sec. calculate the rate constant, k.
$0.217 \text{ sec}^{-1}$
$0.313 \text{ sec}^{-1}$
$8.88 \times 10^{-4} \text{ sec}^{-1}$
$0.150 \text{ sec}^{-1}$
Step1: Recall first-order half-life formula
For a first-order reaction, the half-life $t_{1/2}$ is related to the rate constant $k$ by:
$$t_{1/2} = \frac{\ln 2}{k}$$
Rearranged to solve for $k$:
$$k = \frac{\ln 2}{t_{1/2}}$$
Step2: Substitute given values
Given $t_{1/2} = 7.80 \times 10^2$ sec, and $\ln 2 \approx 0.6931$:
$$k = \frac{0.6931}{7.80 \times 10^2}$$
Step3: Calculate the value
$$k = \frac{0.6931}{780} \approx 8.88 \times 10^{-4} \text{ sec}^{-1}$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
8.88 x 10⁻⁴ sec⁻¹