QUESTION IMAGE
Question
having problems staying logged in or are you experiencing issues? please visit our troubleshooting section for solutions
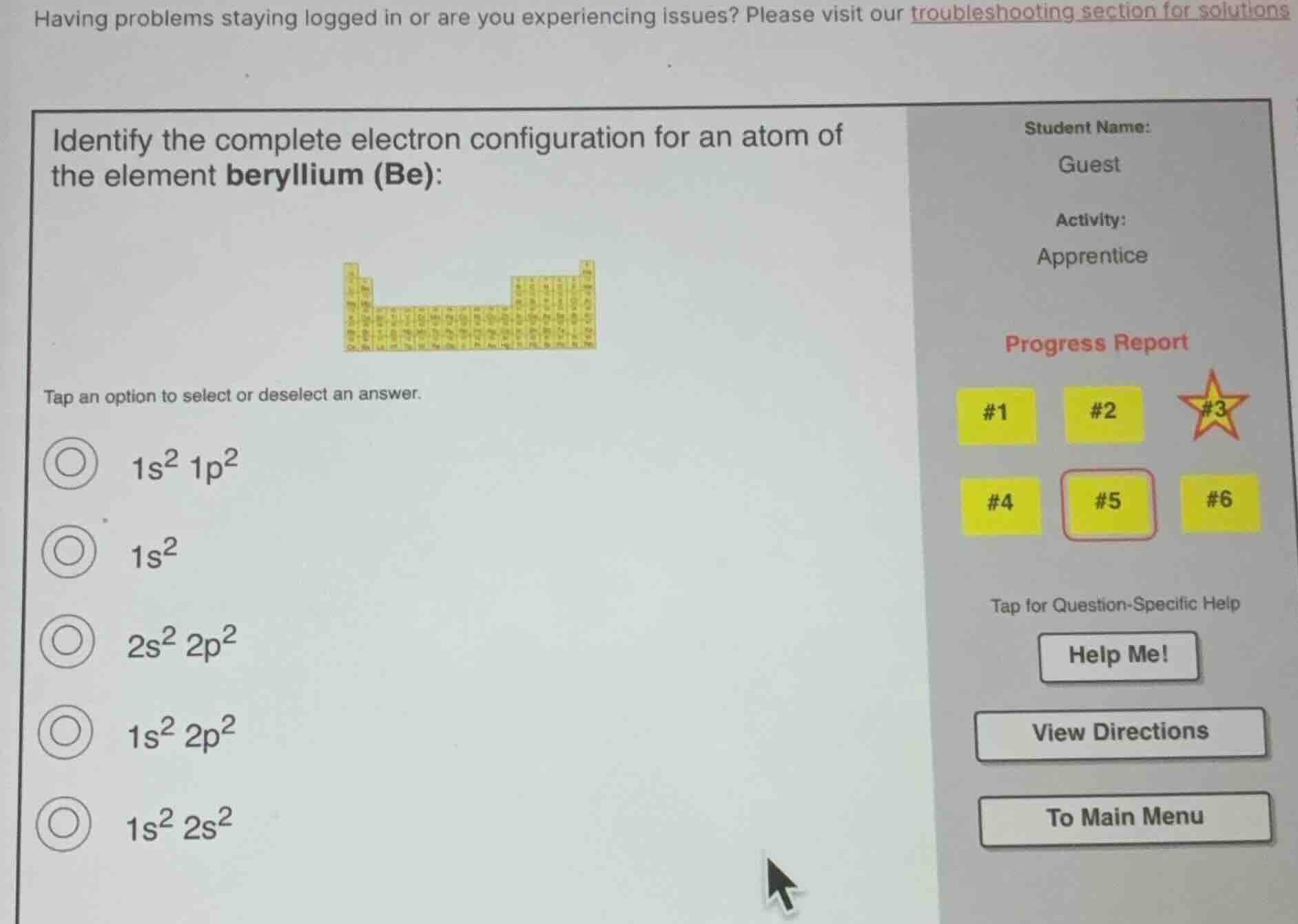
identify the complete electron configuration for an atom of
the element beryllium (be):
tap an option to select or deselect an answer.
$1s^{2}1p^{2}$
$1s^{2}$
$2s^{2}2p^{2}$
$1s^{2}2p^{2}$
$1s^{2}2s^{2}$
student name:
guest
activity:
apprentice
progress report
#1 #2 #3
#4 #5 #6
tap for question-specific help
help me!
view directions
to main menu
Step1: Find Be's atomic number
Beryllium (Be) has an atomic number of 4, so it has 4 electrons.
Step2: Follow Aufbau principle
Fill orbitals in order: 1s, 2s, 2p... The 1s orbital holds max 2 electrons, then fill 2s.
$$1s^2 2s^2$$
This accounts for all 4 electrons. Eliminate options with non-existent $1p$ orbitals, or incomplete/incorrect orbital filling.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1s² 2s²