QUESTION IMAGE
Question
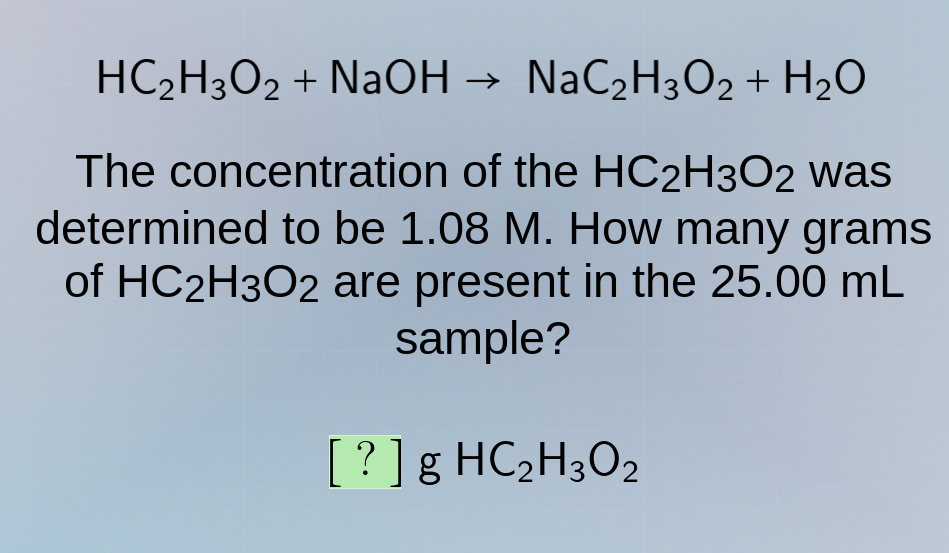
hc₂h₃o₂ + naoh → nac₂h₃o₂ + h₂o
the concentration of the hc₂h₃o₂ was determined to be 1.08 m. how many grams of hc₂h₃o₂ are present in the 25.00 ml sample?
? g hc₂h₃o₂
Step1: Convert volume to liters
The volume of the solution is 25.00 mL. Since 1 L = 1000 mL, we convert mL to L by dividing by 1000.
\( V = 25.00\space mL\times\frac{1\space L}{1000\space mL}= 0.025\space L \)
Step2: Calculate moles of \( HC_2H_3O_2 \)
Molarity (M) is moles of solute per liter of solution. The formula is \( M=\frac{n}{V} \), where \( n \) is moles and \( V \) is volume in liters. Rearranging for \( n \), we get \( n = M\times V \).
Given \( M = 1.08\space M \) and \( V = 0.025\space L \),
\( n = 1.08\space mol/L\times0.025\space L = 0.027\space mol \)
Step3: Calculate molar mass of \( HC_2H_3O_2 \) (acetic acid)
The molar mass of \( H = 1.008\space g/mol \), \( C = 12.01\space g/mol \), \( O = 16.00\space g/mol \).
For \( HC_2H_3O_2 \) (which is \( C_2H_4O_2 \)):
\( Molar\space mass = 2\times12.01 + 4\times1.008 + 2\times16.00 \)
\( = 24.02 + 4.032 + 32.00 = 60.052\space g/mol \)
Step4: Calculate mass of \( HC_2H_3O_2 \)
Mass \( m = n\times Molar\space mass \)
\( m = 0.027\space mol\times60.052\space g/mol \approx 1.6214\space g \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 1.62 \) (or more precisely \( 1.6214 \))