QUESTION IMAGE
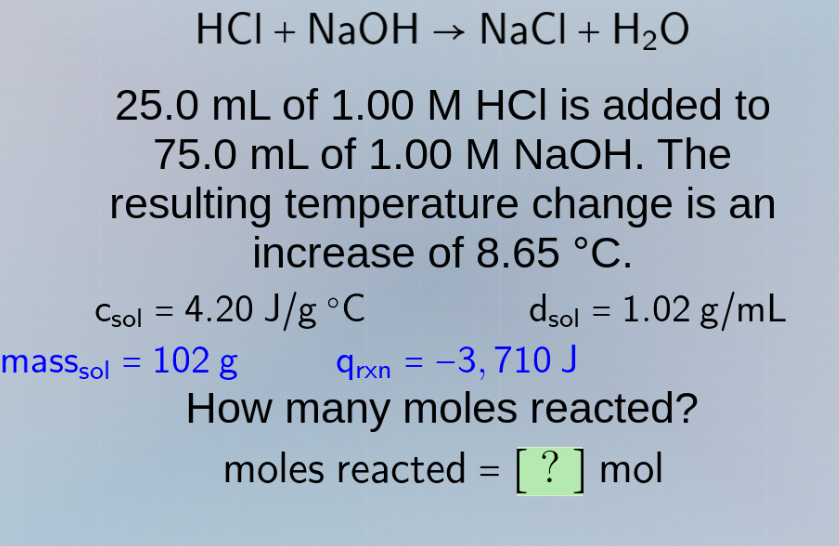
Question
hcl + naoh → nacl + h₂o
25.0 ml of 1.00 m hcl is added to
75.0 ml of 1.00 m naoh. the
resulting temperature change is an
increase of 8.65 °c.
csol = 4.20 j/g °c dsol = 1.02 g/ml
masssol = 102 g qrxn = −3,710 j
how many moles reacted?
moles reacted = ? mol
Step1: Determine moles of HCl
Molarity of HCl is \(1.00\ M\) and volume is \(25.0\ mL = 0.025\ L\). Using the formula \(n = M\times V\), moles of HCl \(n_{HCl}=1.00\ mol/L\times0.025\ L = 0.025\ mol\).
Step2: Determine moles of NaOH
Molarity of NaOH is \(1.00\ M\) and volume is \(75.0\ mL = 0.075\ L\). Moles of NaOH \(n_{NaOH}=1.00\ mol/L\times0.075\ L = 0.075\ mol\).
Step3: Identify limiting reactant
The reaction is \(HCl + NaOH
ightarrow NaCl + H_2O\), the mole ratio of \(HCl\) to \(NaOH\) is \(1:1\). Since \(n_{HCl}=0.025\ mol\) and \(n_{NaOH}=0.075\ mol\), \(HCl\) is the limiting reactant, so moles reacted is equal to moles of \(HCl\) (or the moles of the limiting reactant which reacts completely).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(0.025\)