QUESTION IMAGE
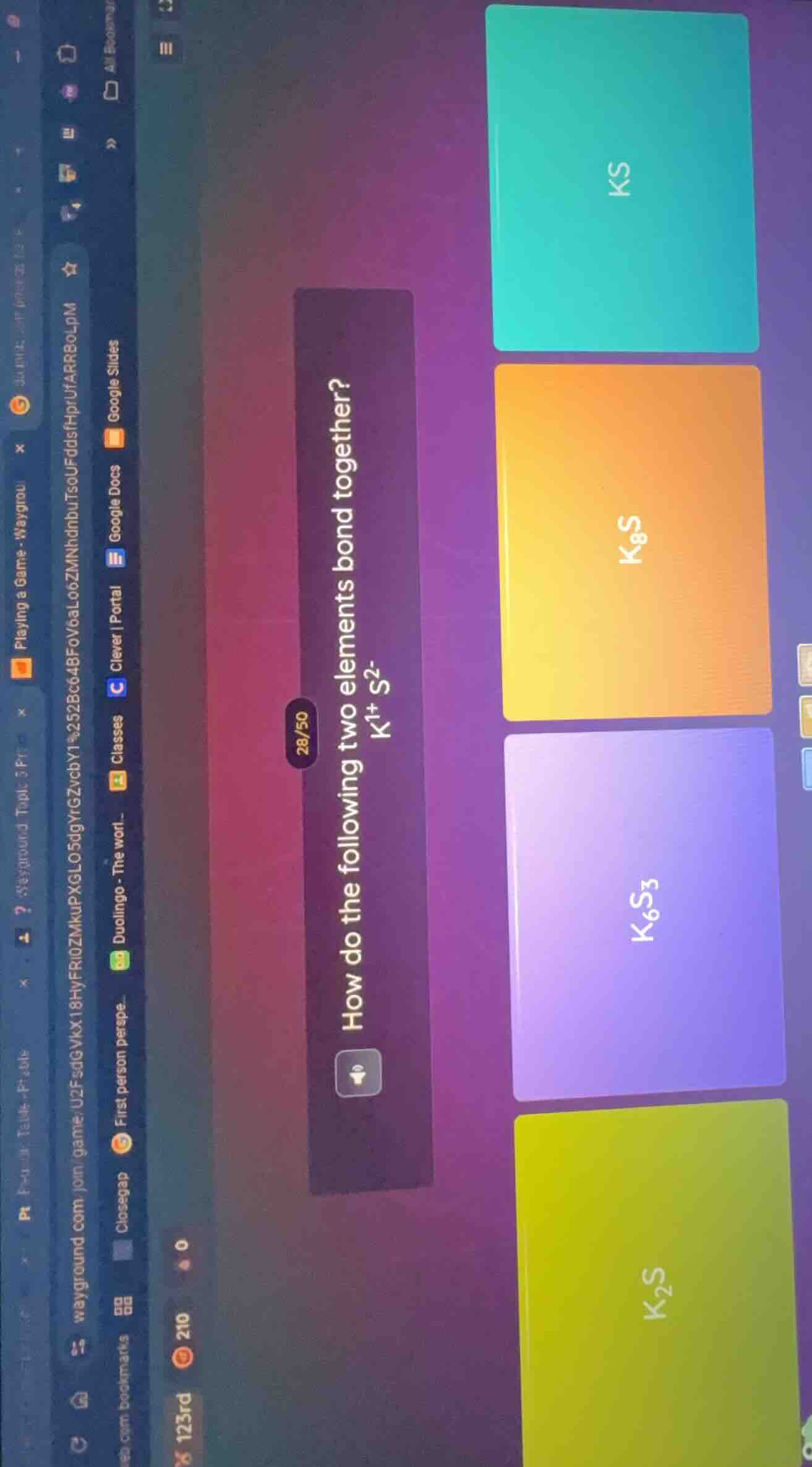
Question
how do the following two elements bond together? k⁺ s²⁻
Step1: Balance ionic charges
Potassium has a +1 charge ($K^+$), sulfur has a -2 charge ($S^{2-}$). We need total positive charge = total negative charge. Let $x$ = number of $K^+$ ions, $y$ = number of $S^{2-}$ ions.
$x(+1) + y(-2) = 0$
Step2: Solve for ion ratio
Substitute to balance: $2(+1) + 1(-2) = 0$. So 2 $K^+$ ions bond with 1 $S^{2-}$ ion.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$K_2S$ (the yellow-green bottom option)