QUESTION IMAGE
Question
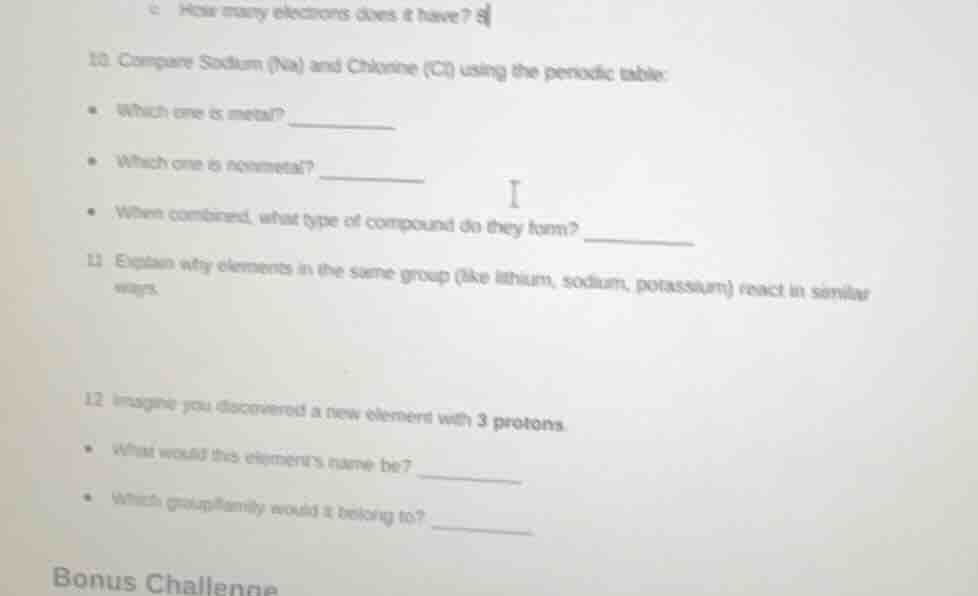
c. how many electrons does it have?
- compare sodium (na) and chlorine (cl) using the periodic table:
- which one is metal?
- which one is nonmetal?
- when combined, what type of compound do they form?
- explain why elements in the same group (like lithium, sodium, potassium) react in similar ways.
- imagine you discovered a new element with 3 protons.
- what would this element’s name be?
- which group/family would it belong to?
bonus challenge
Question 10
Part 1: Which one is metal?
Metals are typically found on the left side and middle of the periodic table. Sodium (Na) is in Group 1 (alkali metals), which are metals. Chlorine (Cl) is in Group 17 (halogens), non - metals.
Non - metals are often on the right side of the periodic table (except for hydrogen). Chlorine (Cl) is a halogen in Group 17, characterized as a non - metal, while Na is a metal.
Sodium (a metal) and Chlorine (a non - metal) react to form an ionic compound. Metals tend to lose electrons and non - metals gain electrons, resulting in the formation of ions that are held together by ionic bonds. The compound formed is sodium chloride (NaCl), an ionic compound.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Sodium (Na)