QUESTION IMAGE
Question
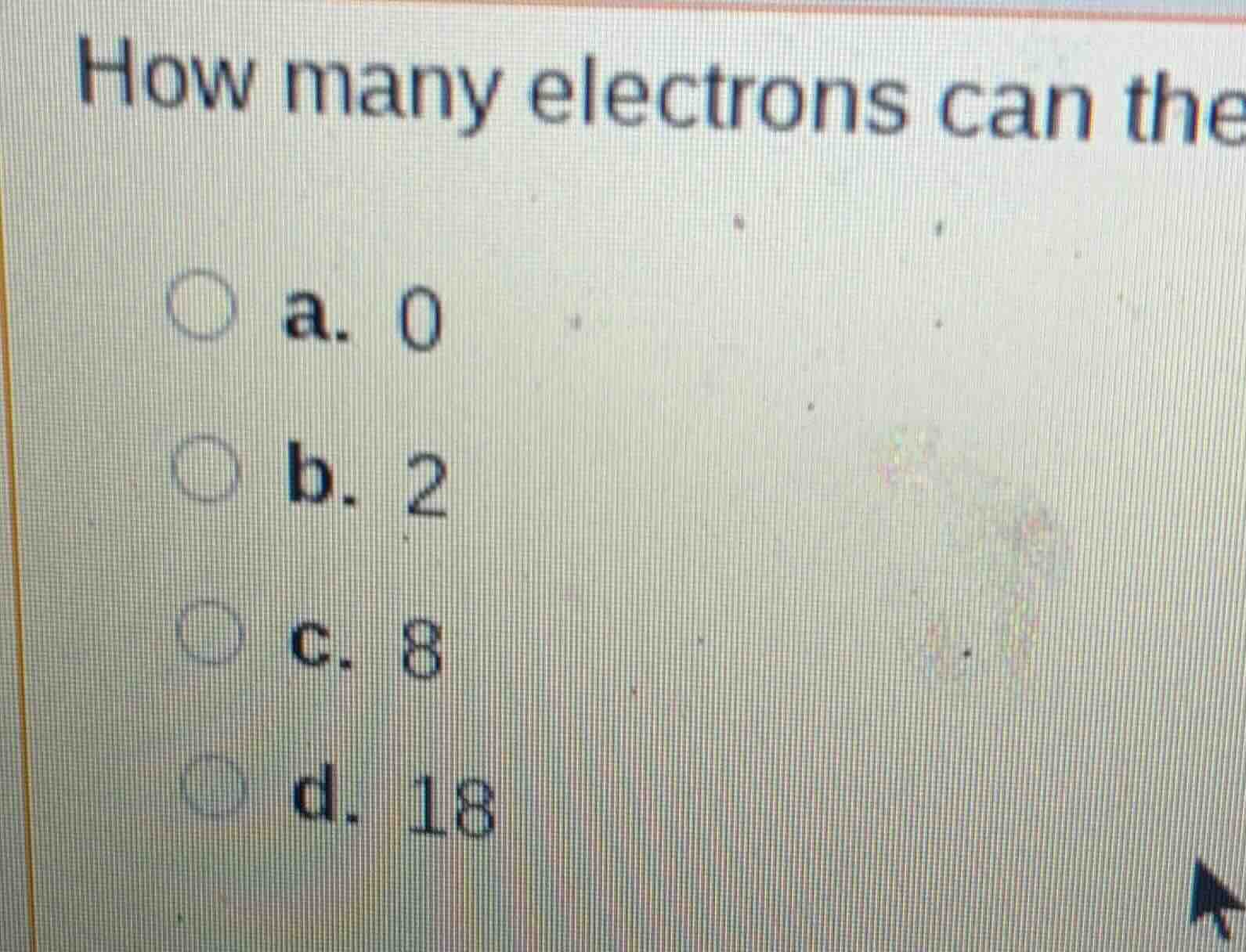
how many electrons can the
a. 0
b. 2
c. 8
d. 18
Brief Explanations
Assuming the question is about the maximum number of electrons in the first electron shell (K shell), the formula for the maximum number of electrons in a shell is $2n^2$, where $n$ is the shell number. For the first shell ($n = 1$), we calculate $2\times(1)^2 = 2$. So the first electron shell can hold a maximum of 2 electrons.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
b. 2