QUESTION IMAGE
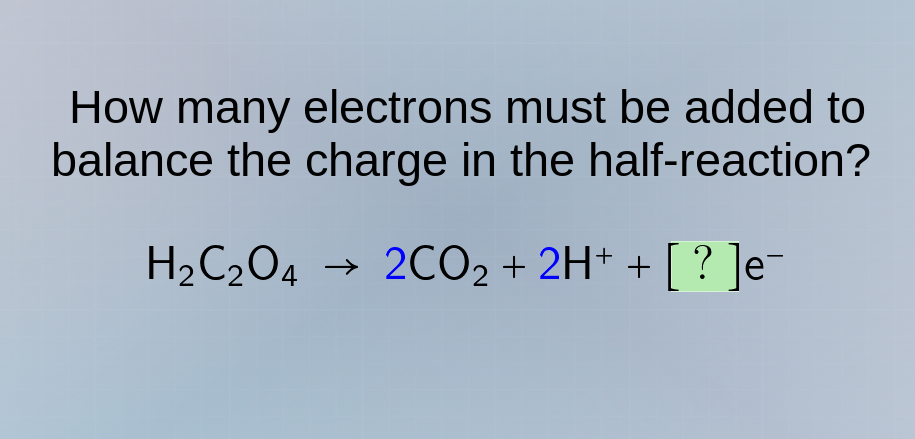
Question
how many electrons must be added to balance the charge in the half - reaction? \\(\ce{h_{2}c_{2}o_{4} \to 2co_{2} + 2h^{+} + ?e^{-}}\\)
Step1: Determine oxidation states of C
In \(H_2C_2O_4\), H is \(+1\), O is \(-2\). Let C be \(x\). \(2(+1)+2x + 4(-2)=0\) → \(2 + 2x-8 = 0\) → \(2x=6\) → \(x = +3\). In \(CO_2\), O is \(-2\), let C be \(y\). \(y+2(-2)=0\) → \(y = +4\). Each C atom loses 1 electron, 2 C atoms lose \(2\times1 = 2\) electrons.
Step2: Check charge balance
Left side (\(H_2C_2O_4\)): charge is 0. Right side: \(2CO_2\) (0) + \(2H^+\) (\(+2\)) + \(ne^-\) (\(-n\)). So \(0=+2 - n\) → \(n = 2\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
2