QUESTION IMAGE
Question
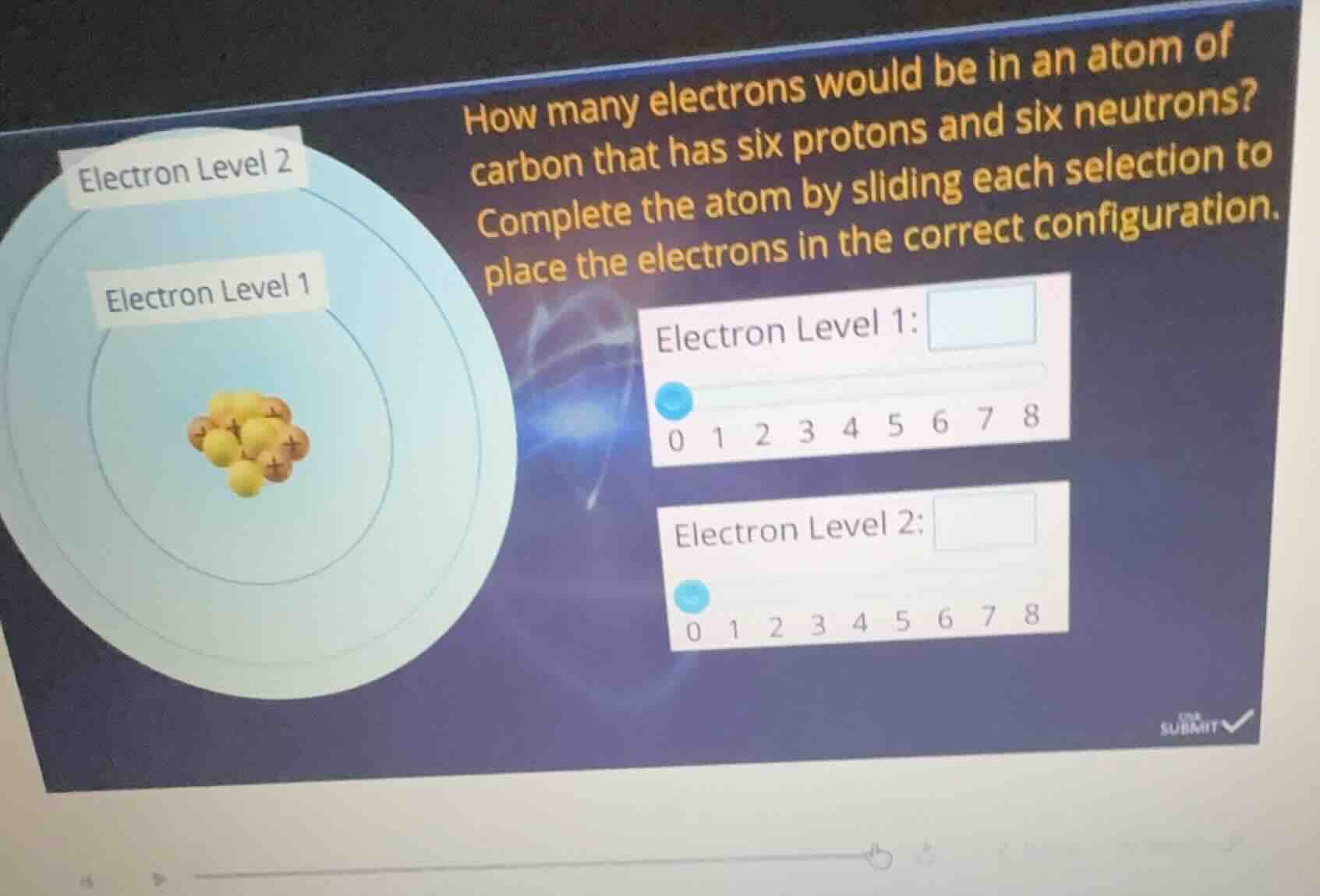
how many electrons would be in an atom of carbon that has six protons and six neutrons? complete the atom by sliding each selection to place the electrons in the correct configuration. electron level 1: 0 1 2 3 4 5 6 7 8 electron level 2: 0 1 2 3 4 5 6 7 8
Step1: Recall atom charge balance
In a neutral atom, number of electrons = number of protons. Given carbon has 6 protons, so total electrons = 6.
Step2: Determine electron level 1 capacity
Electron level 1 (K - shell) can hold a maximum of 2 electrons.
Step3: Calculate electrons in level 2
Subtract electrons in level 1 from total electrons: \( 6 - 2 = 4 \). So level 1 has 2, level 2 has 4.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Electron Level 1: 2
Electron Level 2: 4