QUESTION IMAGE
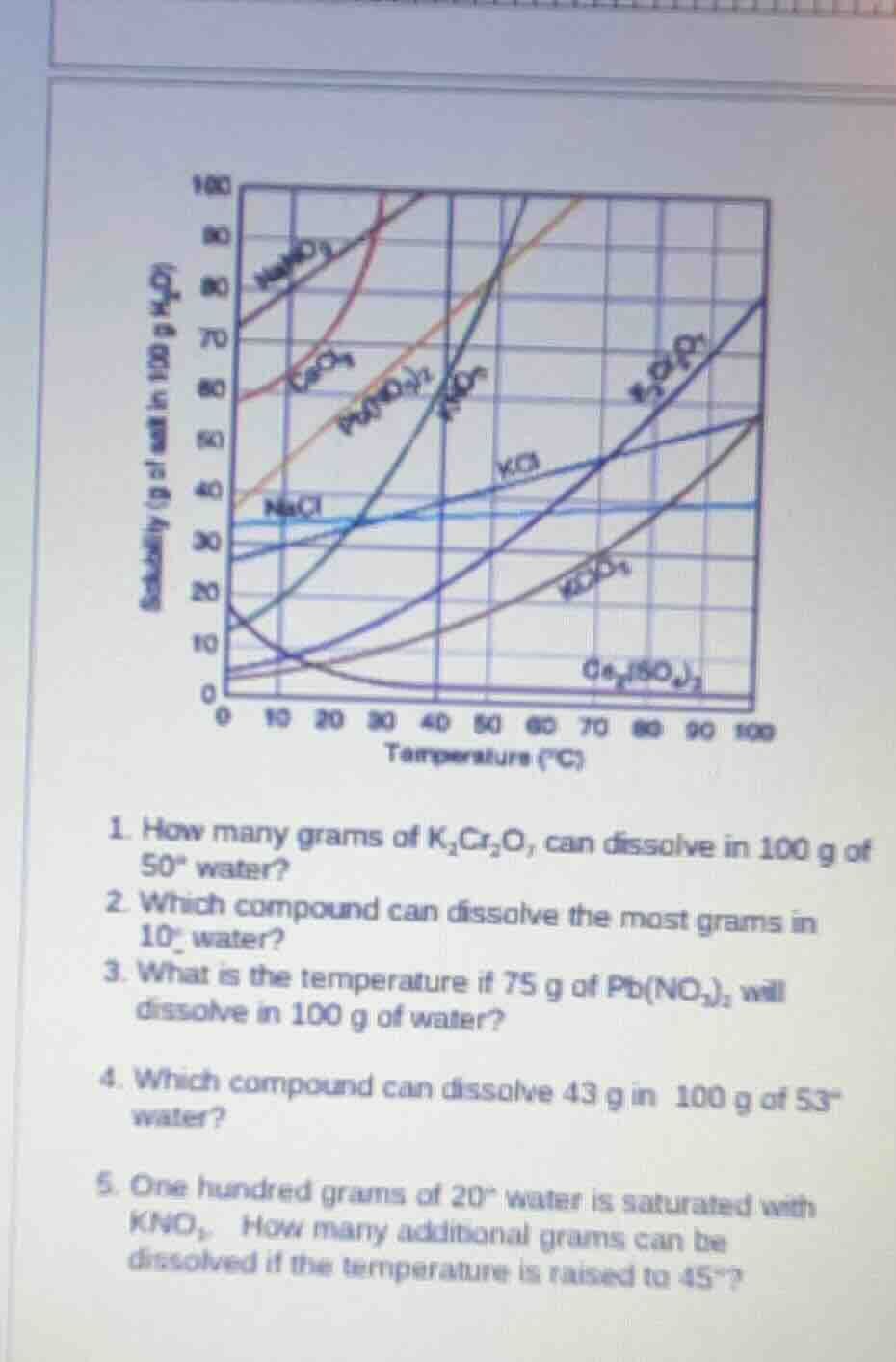
Question
- how many grams of k₂cr₂o₇ can dissolve in 100 g of 50° water?
- which compound can dissolve the most grams in 10° water?
- what is the temperature if 75 g of pb(no₃)₂ will dissolve in 100 g of water?
- which compound can dissolve 43 g in 100 g of 53° water?
- one hundred grams of 20° water is saturated with kno₃. how many additional grams can be dissolved if the temperature is raised to 45°?
Question 1:
Step1: Locate the solubility curve for \(K_2Cr_2O_7\) and find the value at \(50^\circ C\) on the temperature axis.
From the solubility curve, at \(50^\circ C\), the solubility of \(K_2Cr_2O_7\) in 100 g of water is found by reading the y - value (grams of solute) corresponding to \(x = 50^\circ C\) on the \(K_2Cr_2O_7\) curve.
Step2: Determine the solubility.
Looking at the curve, the solubility of \(K_2Cr_2O_7\) at \(50^\circ C\) in 100 g of water is approximately 25 g (this value is obtained from typical solubility curves for \(K_2Cr_2O_7\); the exact value may vary slightly depending on the curve, but around 25 g is a common value).
Step1: Locate the solubility curves of all compounds at \(10^\circ C\).
We look at the y - axis (grams of solute in 100 g of water) for each compound's curve at \(x = 10^\circ C\).
Step2: Compare the solubilities.
Compounds like \(NaNO_3\) have a relatively high solubility at lower temperatures. By comparing the y - values of different compound curves at \(10^\circ C\), we find that \(NaNO_3\) has the highest solubility (the solubility of \(NaNO_3\) at \(10^\circ C\) is relatively higher than other compounds like \(KNO_3\), \(NaCl\), etc. at this temperature).
To determine which compound dissolves the most in \(10^\circ\) water, we analyze the solubility curves at \(10^\circ C\). By comparing the amount of solute (in grams per 100 g of water) for each compound at \(10^\circ C\), we find that \(NaNO_3\) has the highest solubility among the given compounds at this temperature.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Approximately 25 grams (the value may vary slightly depending on the exact solubility curve, but around 25 g is typical).