QUESTION IMAGE
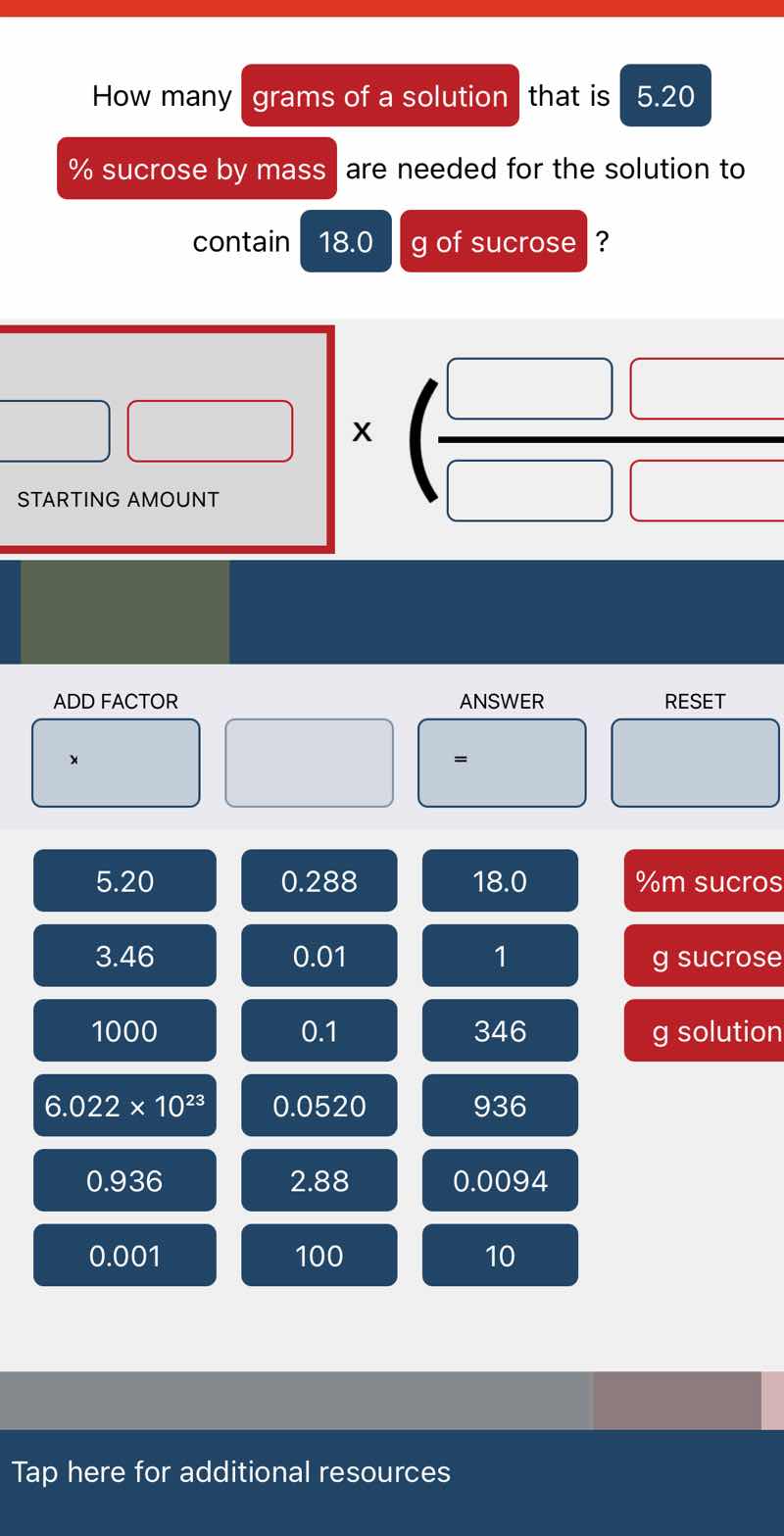
Question
how many grams of a solution that is 5.20 % sucrose by mass are needed for the solution to contain 18.0 g of sucrose?
Step1: Define mass percent formula
Mass percent: $\text{Mass \%} = \frac{\text{Mass of solute}}{\text{Mass of solution}} \times 100\%$
Rearrange to solve for mass of solution:
$\text{Mass of solution} = \frac{\text{Mass of solute}}{\text{Mass \%}} \times 100\%$
Step2: Substitute given values
Given: Mass of solute (sucrose) = $18.0\ \text{g}$, Mass % = $5.20\%$
$\text{Mass of solution} = \frac{18.0}{5.20} \times 100$
Step3: Calculate the result
$\frac{18.0}{5.20} \times 100 \approx 346$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
346 g solution