QUESTION IMAGE
Question
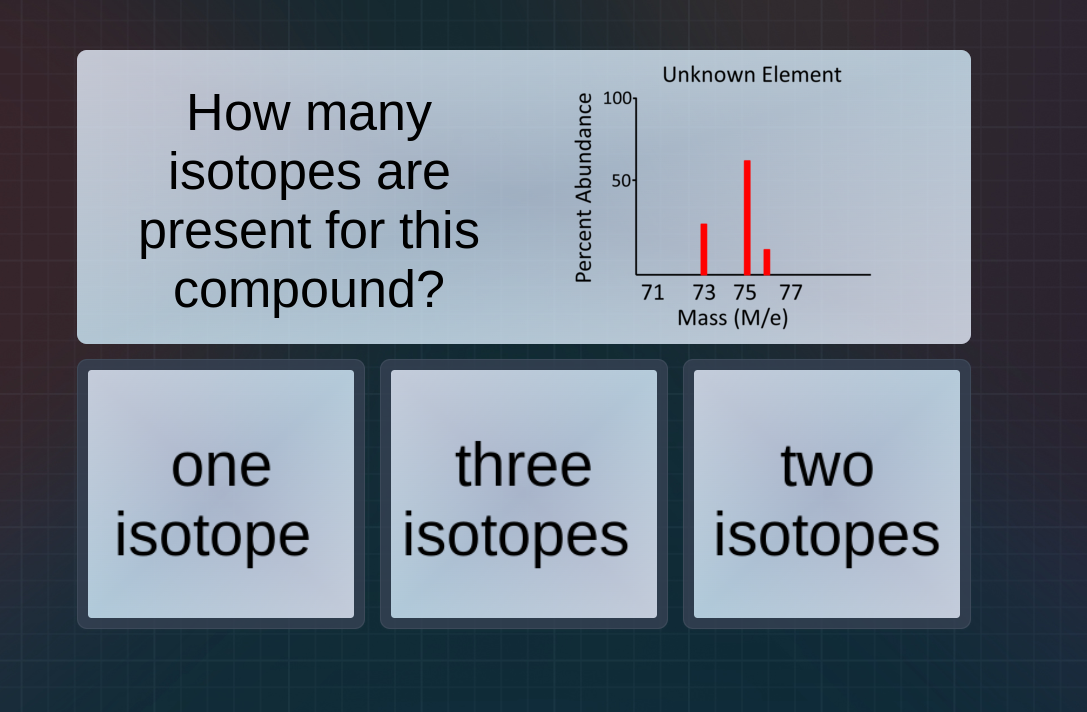
how many isotopes are present for this compound? unknown element percent abundance 100 50 71 73 75 77 mass (m/e) one isotope three isotopes two isotopes
To determine the number of isotopes, we analyze the mass spectrum. Each peak in the mass spectrum (with mass values 73, 75, and the small one around 77? Wait, no, looking at the x - axis: 73, 75, and another? Wait, the x - axis labels are 71, 73, 75, 77. The peaks are at 73, 75, and a small one (maybe 77? Or is there a peak at 73, 75, and another? Wait, the graph has three peaks (at 73, 75, and a small one, maybe 77? Wait, no, the first peak is at 73, second at 75, third at a value after 75, maybe 77. Wait, the number of peaks in the mass spectrum for isotopes corresponds to the number of isotopes. Each peak represents an isotope with a different mass. Looking at the mass (m/e) axis, the peaks are at 73, 75, and another (the small one). Wait, no, the x - axis has 71, 73, 75, 77. The peaks are at 73, 75, and a third peak (maybe at 77? Or is the first peak at 73, second at 75, third at a value between 75 and 77? Wait, the key is that each peak in the mass spectrum (for isotopic abundance) represents an isotope. So if there are three peaks, there are three isotopes. Wait, looking at the options: one, three, two. The peaks are at 73, 75, and a third (the small one). So three peaks mean three isotopes.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. three isotopes