QUESTION IMAGE
Question
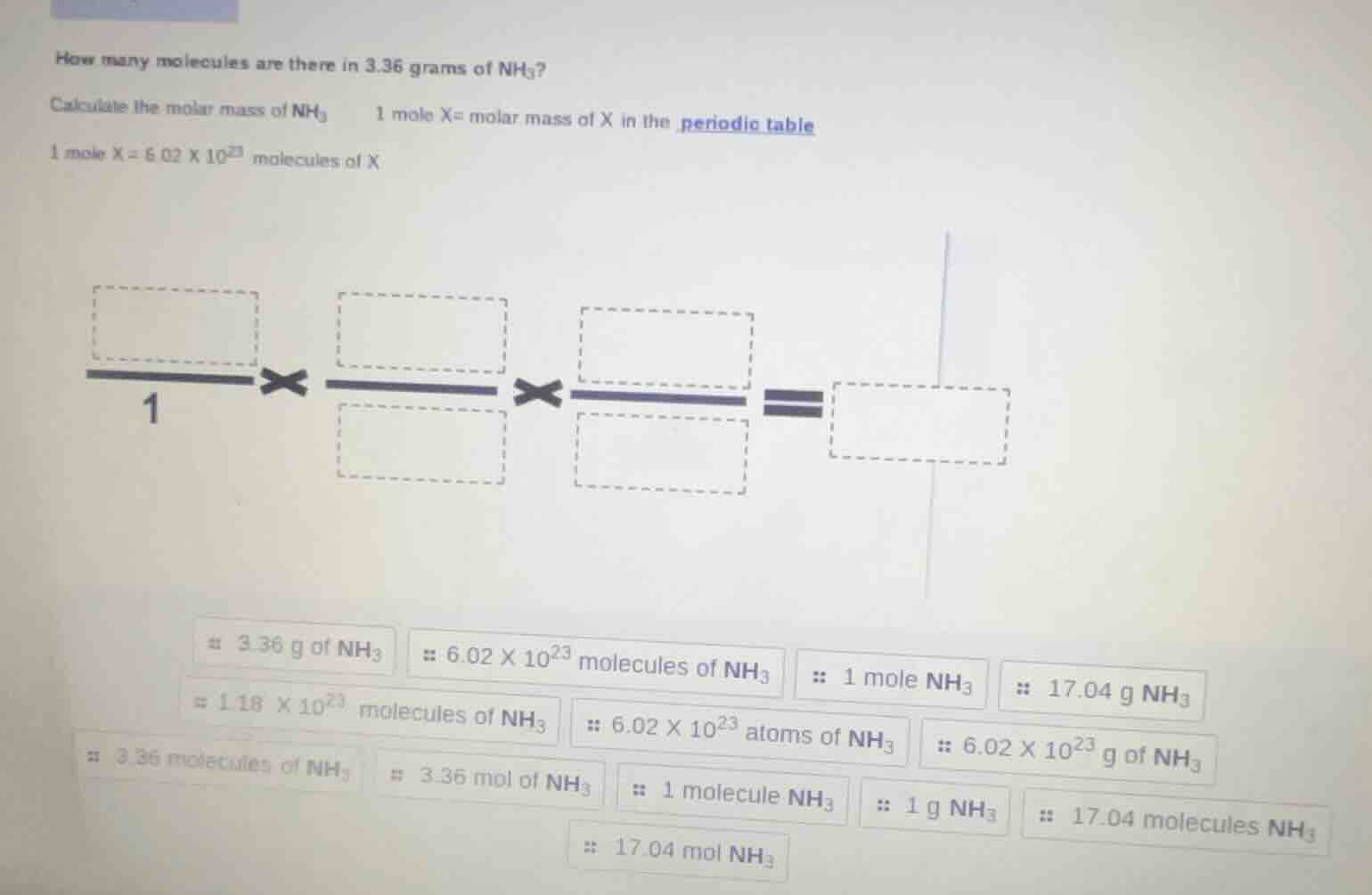
how many molecules are there in 3.36 grams of $nh_3$?
calculate the molar mass of $nh_3$ \quad 1 mole x= molar mass of x in the periodic table
1 mole x = $6.02 \times 10^{23}$ molecules of x
Step1: Calculate molar mass of \( NH_3 \)
Molar mass of \( N \) is \( 14.01 \, g/mol \), molar mass of \( H \) is \( 1.01 \, g/mol \). For \( NH_3 \), molar mass \( = 14.01 + 3\times1.01 = 17.04 \, g/mol \).
Step2: Convert grams to moles
Moles of \( NH_3 = \frac{3.36 \, g}{17.04 \, g/mol} \)
Step3: Convert moles to molecules
Molecules of \( NH_3 = \) Moles \( \times 6.02 \times 10^{23} \)
Putting it together: \( \frac{3.36 \, g}{17.04 \, g/mol} \times 6.02 \times 10^{23} \, molecules/mol \)
Calculate: \( \frac{3.36}{17.04} \approx 0.1972 \); \( 0.1972 \times 6.02 \times 10^{23} \approx 1.18 \times 10^{23} \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 1.18 \times 10^{23} \) molecules of \( NH_3 \)