QUESTION IMAGE
Question
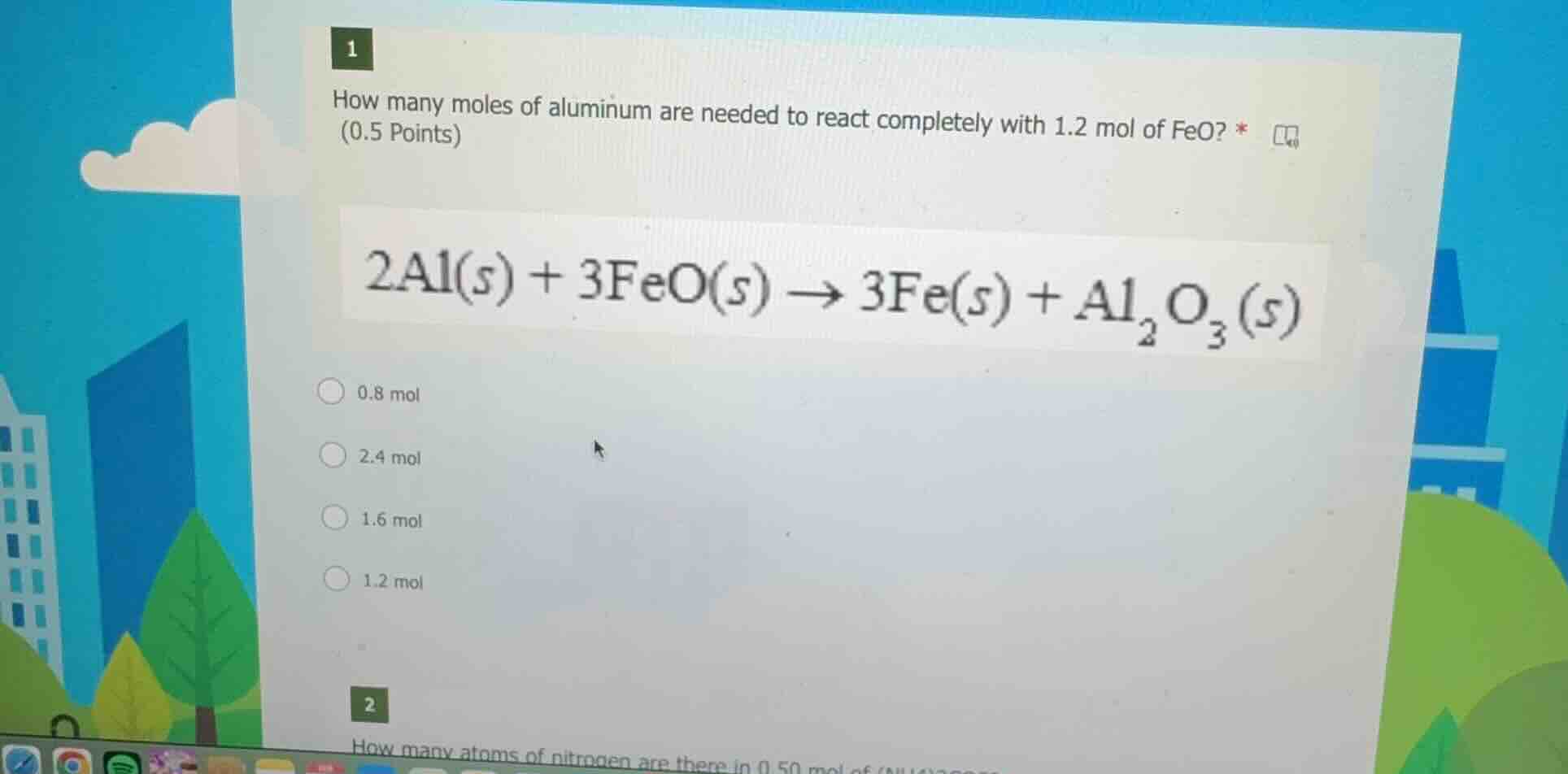
1
how many moles of aluminium are needed to react completely with 1.2 mol of feo? *
(0.5 points)
2al(s) + 3feo(s) → 3fe(s) + al₂o₃(s)
0.8 mol
2.4 mol
1.6 mol
1.2 mol
2
how many atoms of nitrogen are there in 0.50 mol of (nh
Step1: Identify mole ratio
From the reaction \( 2\text{Al}(s) + 3\text{FeO}(s)
ightarrow 3\text{Fe}(s) + \text{Al}_2\text{O}_3(s) \), the mole ratio of \(\text{Al}\) to \(\text{FeO}\) is \( \frac{2}{3} \).
Step2: Calculate moles of Al
Let \( n_{\text{Al}} \) be moles of Al. Using the ratio: \( n_{\text{Al}} = 1.2\ \text{mol FeO} \times \frac{2\ \text{mol Al}}{3\ \text{mol FeO}} \)
\( n_{\text{Al}} = \frac{1.2 \times 2}{3} = 0.8\ \text{mol} \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
0.8 mol