QUESTION IMAGE
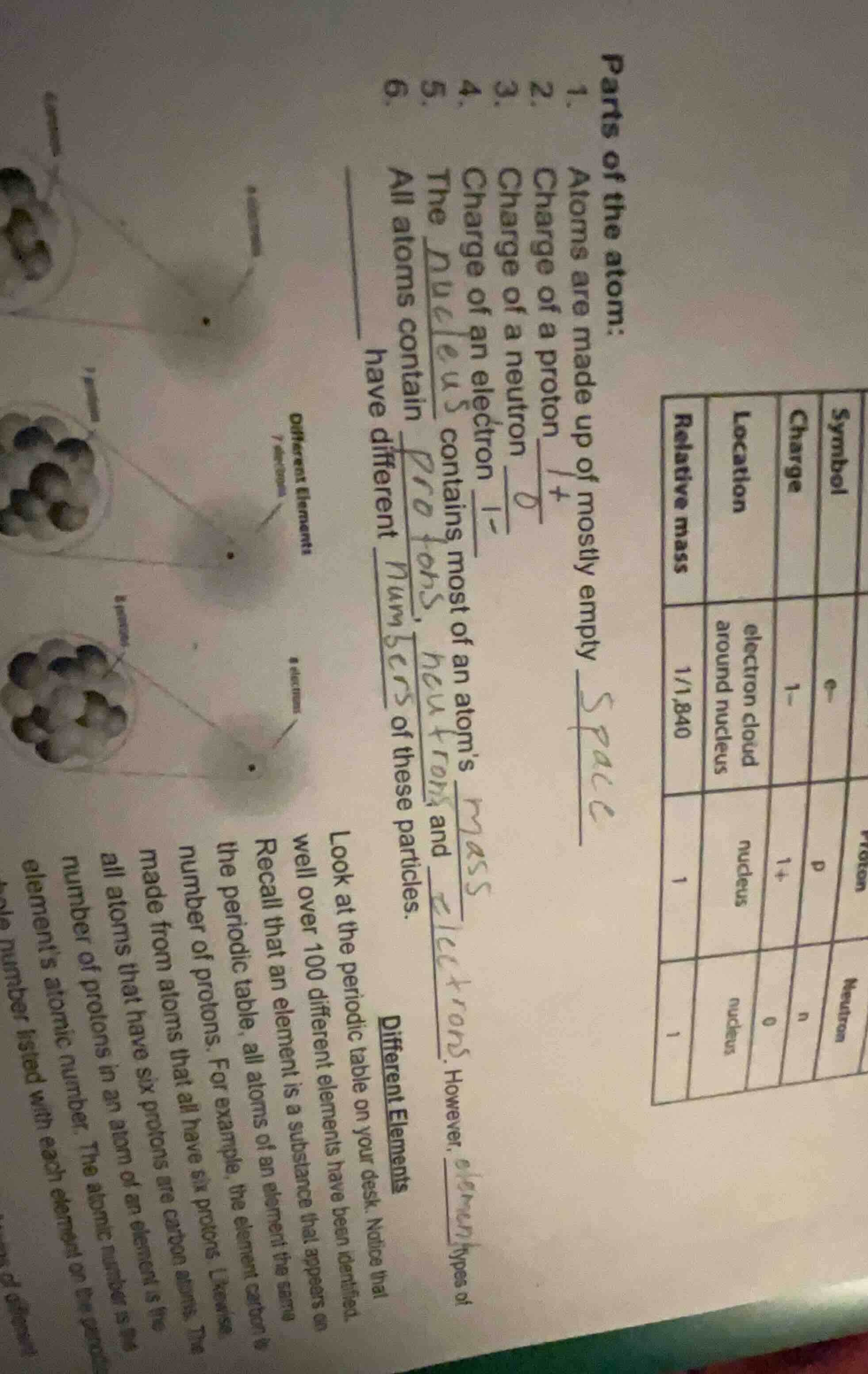
Question
Question was provided via image upload.
Step1: Fill in atom structure blanks
- Atoms are made up of mostly empty space
- Charge of a proton: $\boldsymbol{1+}$
- Charge of a neutron: $\boldsymbol{0}$
- Charge of an electron: $\boldsymbol{1-}$
- The nucleus contains most of an atom's mass
- All atoms contain protons, neutrons, and electrons. However, different elements have different numbers of these particles.
Step2: Verify table completeness
The provided table is already fully completed correctly:
| Particle | Symbol | Charge | Location | Relative mass |
|---|---|---|---|---|
| Proton | $\text{p}$ | $1+$ | nucleus | $1$ |
| Neutron | $\text{n}$ | $0$ | nucleus | $1$ |
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- space
- $\boldsymbol{1+}$
- $\boldsymbol{0}$
- $\boldsymbol{1-}$
- nucleus; mass
- protons, neutrons, and electrons; different elements; numbers
The provided table is already correctly filled out as shown above.