QUESTION IMAGE
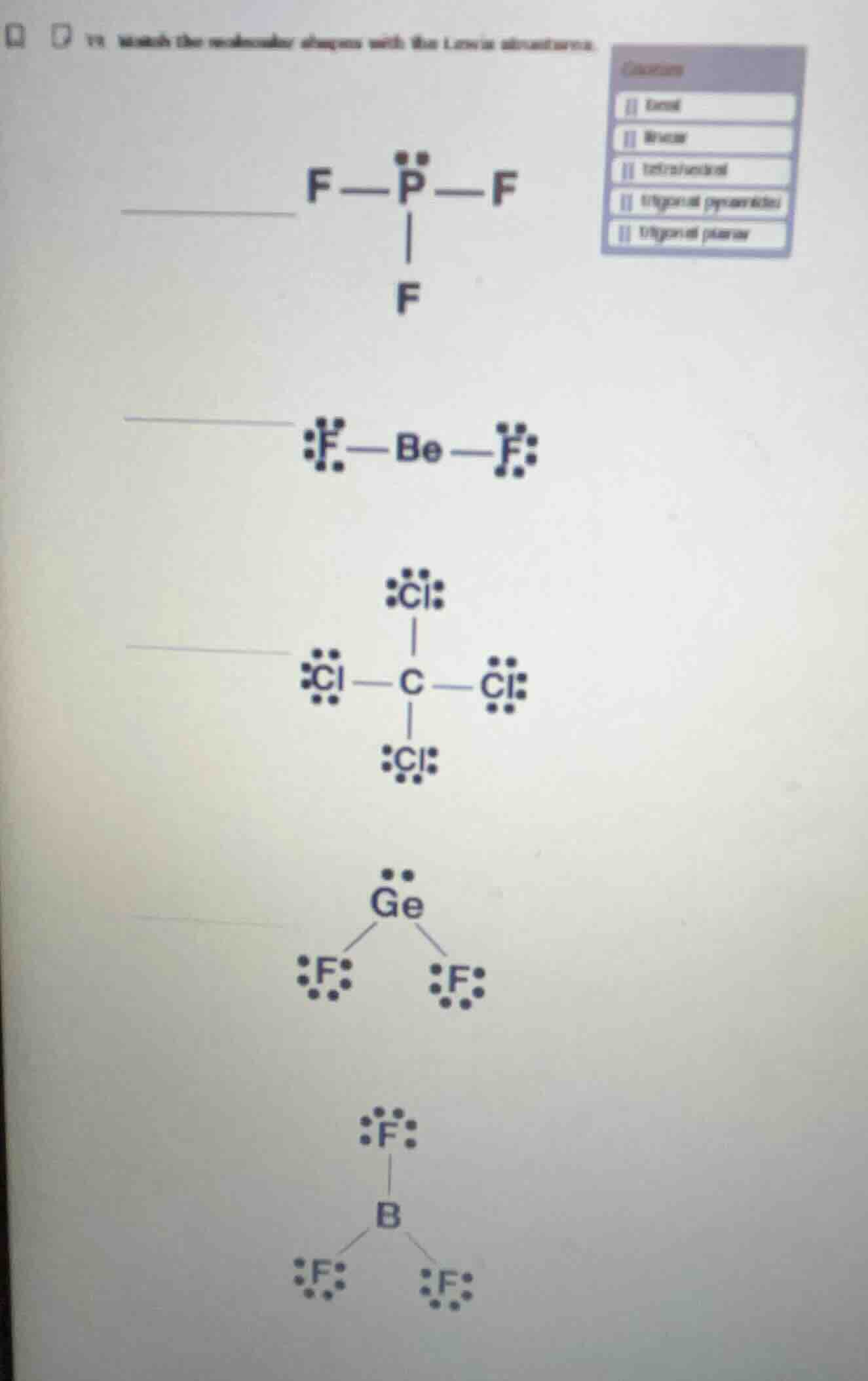
Question
Question was provided via image upload.
Step1: Analyze $\text{PF}_3$
Central P has 3 bonding pairs + 1 lone pair. Electron domain geometry is tetrahedral, molecular geometry is trigonal pyramidal.
Step2: Analyze $\text{BeF}_2$
Central Be has 2 bonding pairs + 0 lone pairs. Molecular geometry is linear.
Step3: Analyze $\text{CCl}_4$
Central C has 4 bonding pairs + 0 lone pairs. Molecular geometry is tetrahedral.
Step4: Analyze $\text{GeF}_2$
Central Ge has 2 bonding pairs + 1 lone pair. Molecular geometry is bent.
Step5: Analyze $\text{BF}_3$
Central B has 3 bonding pairs + 0 lone pairs. Molecular geometry is trigonal planar.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\text{PF}_3$ → trigonal pyramidal
- $\text{BeF}_2$ → linear
- $\text{CCl}_4$ → tetrahedral
- $\text{GeF}_2$ → bent
- $\text{BF}_3$ → trigonal planar