QUESTION IMAGE
Question
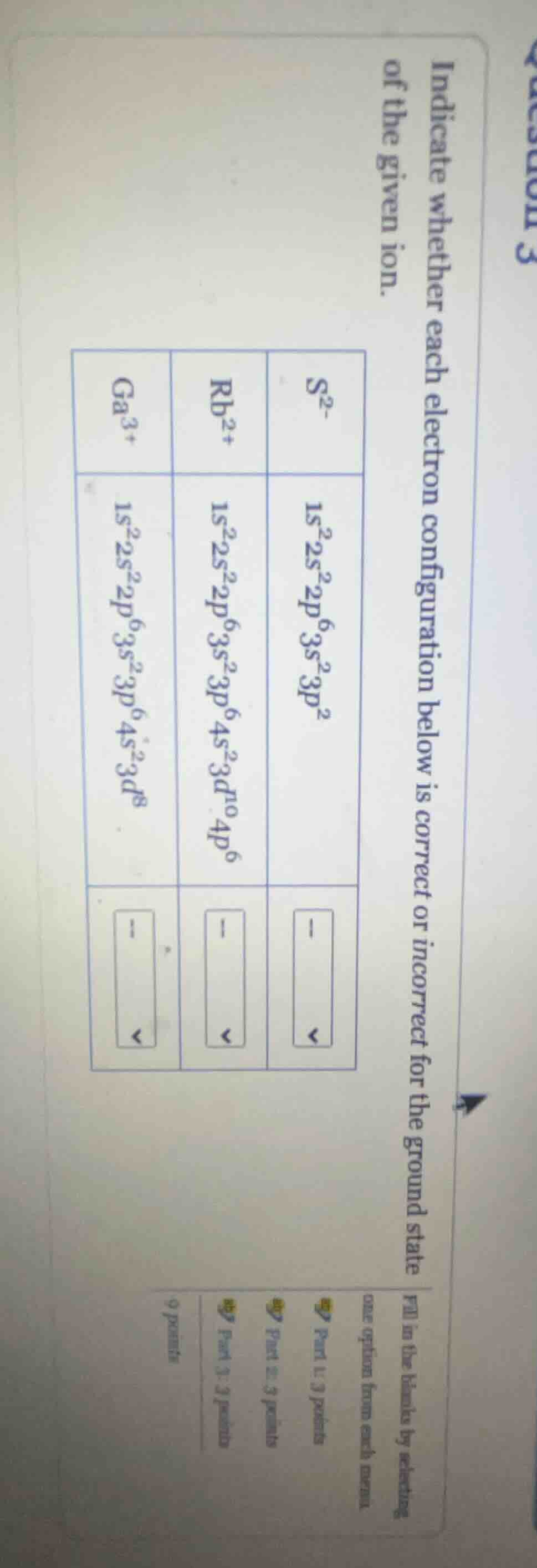
indicate whether each electron configuration below is correct or incorrect for the ground state of the given ion.
s$^{2-}$: $1s^2 2s^2 2p^6 3s^2 3p^2$
rb$^{2+}$: $1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^{10} 4p^6$
ga$^{3+}$: $1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^8$
fill in the blanks by selecting one option from each menu.
part 1: 3 points
part 2: 3 points
part 3: 3 points
9 points
Step1: Check $\text{S}^{2-}$ electron config
Neutral S: $1s^22s^22p^63s^23p^4$. Gain 2 e⁻: $1s^22s^22p^63s^23p^6$. Given config $1s^22s^22p^63s^23p^2$ is wrong.
Step2: Check $\text{Rb}^{2+}$ electron config
Neutral Rb: $1s^22s^22p^63s^23p^64s^23d^{10}4p^65s^1$. Lose 2 e⁻: $1s^22s^22p^63s^23p^64s^23d^{10}4p^5$. Given config $1s^22s^22p^63s^23p^64s^23d^{10}4p^6$ is wrong.
Step3: Check $\text{Ga}^{3+}$ electron config
Neutral Ga: $1s^22s^22p^63s^23p^64s^23d^{10}4p^1$. Lose 3 e⁻: $1s^22s^22p^63s^23p^63d^{10}$. Given config $1s^22s^22p^63s^23p^64s^23d^8$ is wrong.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\text{S}^{2-}$: Incorrect
- $\text{Rb}^{2+}$: Incorrect
- $\text{Ga}^{3+}$: Incorrect