QUESTION IMAGE
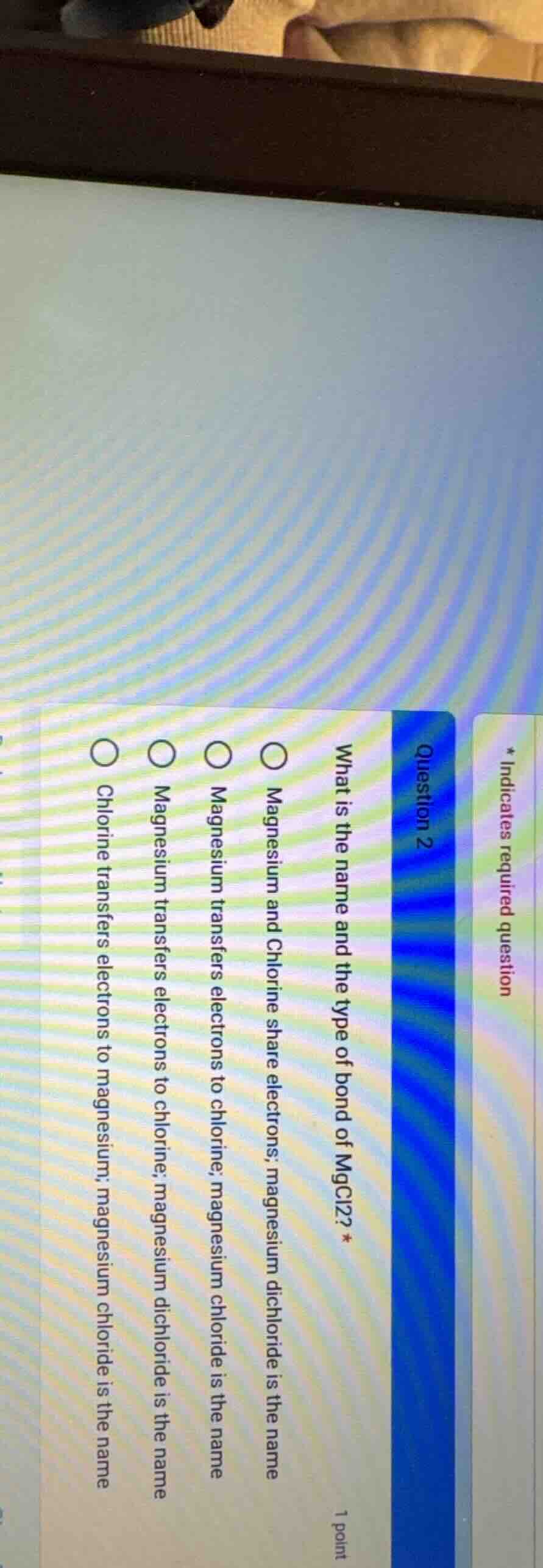
Question
- indicates required question
question 2
what is the name and the type of bond of mgcl₂? *
1 point
○ magnesium and chlorine share electrons; magnesium dichloride is the name
○ magnesium transfers electrons to chlorine; magnesium chloride is the name
○ magnesium transfers electrons to chlorine; magnesium dichloride is the name
○ chlorine transfers electrons to magnesium; magnesium chloride is the name
Magnesium (a group 2 metal) has 2 valence electrons, and chlorine (a group 17 nonmetal) has 7 valence electrons. Magnesium transfers its 2 valence electrons to two separate chlorine atoms (each gains 1 electron) to form an ionic bond. The compound is named magnesium chloride (since magnesium forms only one cation, no prefix is needed for the anion in ionic naming).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Magnesium transfers electrons to chlorine; magnesium chloride is the name