QUESTION IMAGE
Question
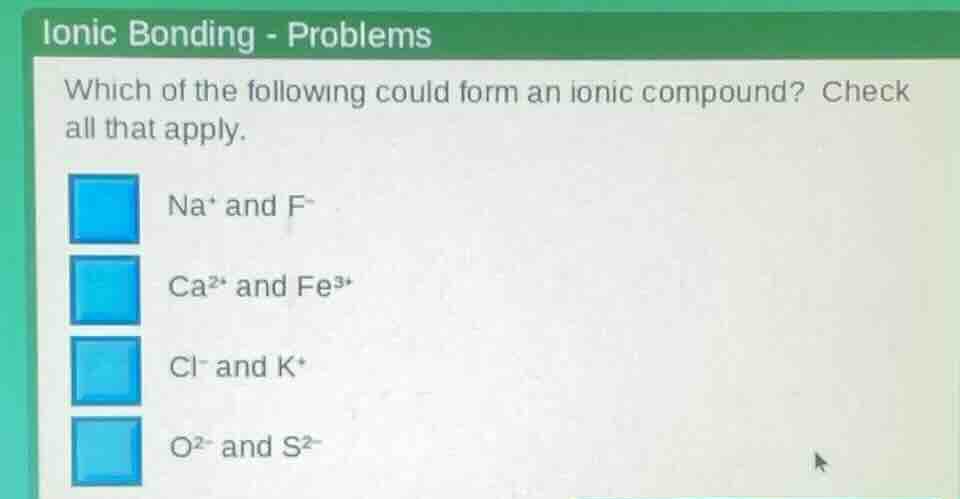
ionic bonding - problems
which of the following could form an ionic compound? check all that apply.
na⁺ and f⁻
ca²⁺ and fe³⁺
cl⁻ and k⁺
o²⁻ and s²⁻
Brief Explanations
Ionic compounds are formed between a cation (positive ion) and an anion (negative ion) due to electrostatic attraction.
- For $\text{Na}^+$ (cation) and $\text{F}^-$ (anion): They are a cation and anion, so they can form an ionic compound (NaF).
- For $\text{Ca}^{2+}$ and $\text{Fe}^{3+}$: Both are cations, so no electrostatic attraction between them to form an ionic compound.
- For $\text{Cl}^-$ (anion) and $\text{K}^+$ (cation): They are an anion and cation, so they can form an ionic compound (KCl).
- For $\text{O}^{2-}$ and $\text{S}^{2-}$: Both are anions, so no electrostatic attraction between them to form an ionic compound.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. $\text{Na}^+$ and $\text{F}^-$
C. $\text{Cl}^-$ and $\text{K}^+$