QUESTION IMAGE
Question
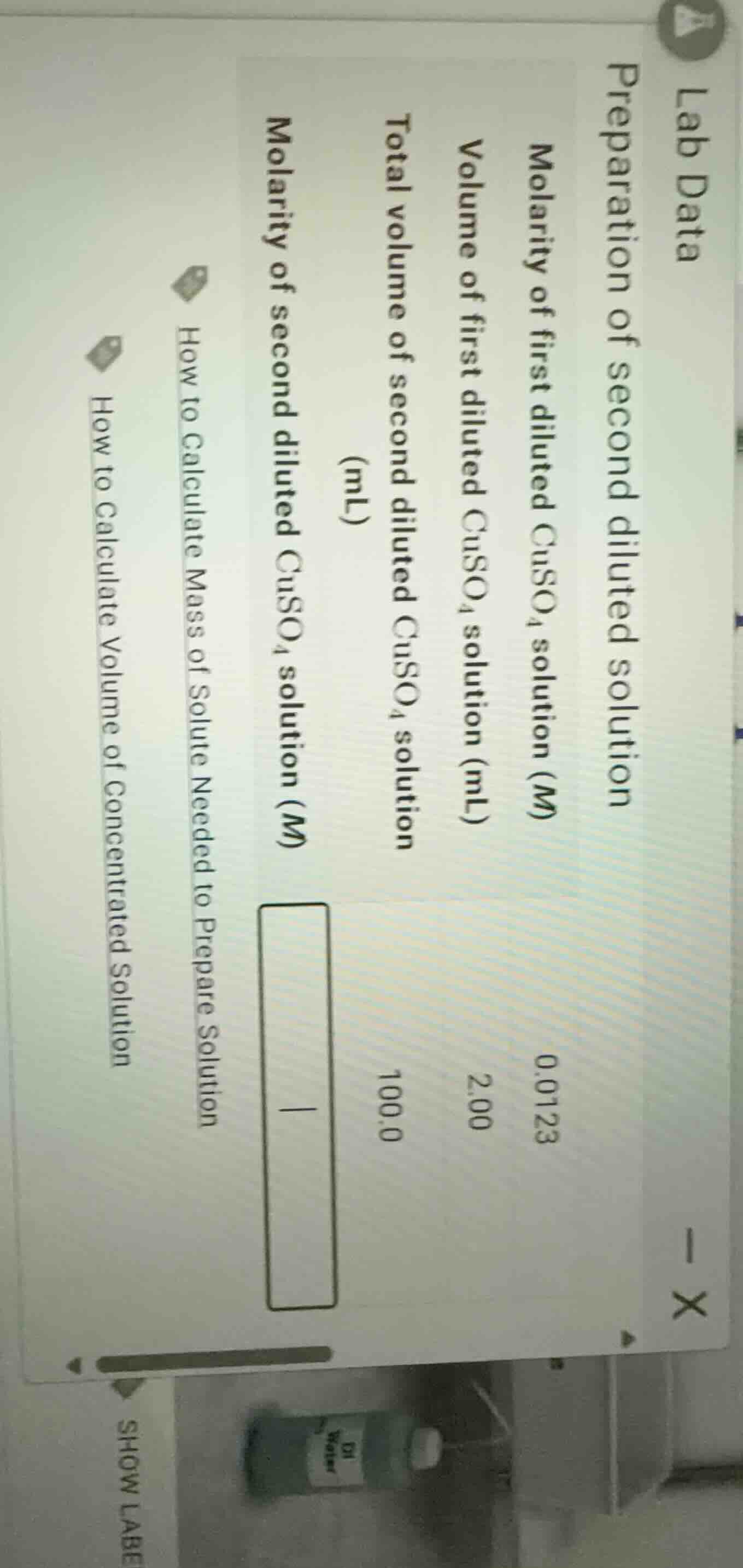
lab data
preparation of second diluted solution
molarity of first diluted $ce{cuso_{4}}$ solution ($m$): 0.0123
volume of first diluted $ce{cuso_{4}}$ solution (ml): 2.00
total volume of second diluted $ce{cuso_{4}}$ solution (ml): 100.0
molarity of second diluted $ce{cuso_{4}}$ solution ($m$)
how to calculate mass of solute needed to prepare solution
how to calculate volume of concentrated solution
Step1: State dilution law formula
$M_1V_1 = M_2V_2$
Step2: Assign values to variables
$M_1=0.0123\ M$, $V_1=2.00\ \text{mL}$, $V_2=100.0\ \text{mL}$
Step3: Rearrange to solve for $M_2$
$M_2 = \frac{M_1V_1}{V_2}$
Step4: Substitute values and calculate
$M_2 = \frac{0.0123 \times 2.00}{100.0}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$0.000246\ M$