QUESTION IMAGE
Question
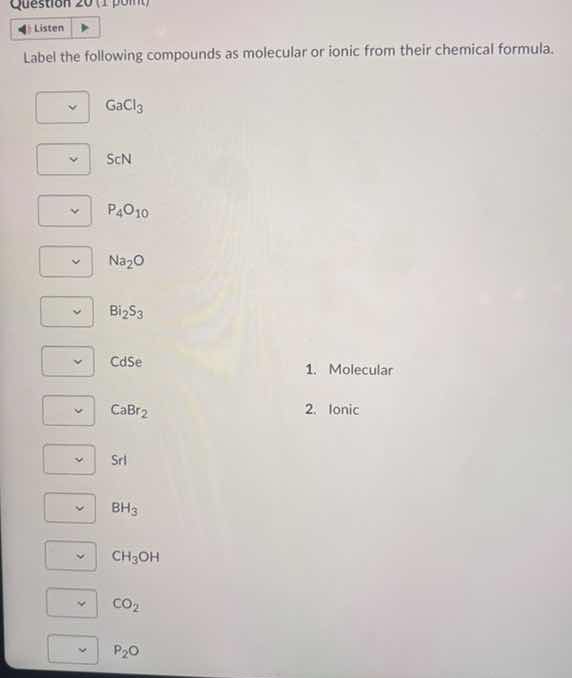
label the following compounds as molecular or ionic from their chemical formula. gacl₃ scn p₄o₁₀ na₂o bi₂s₃ cdse cabr₂ sri bh₃ ch₃oh co₂ p₂o 1. molecular 2. ionic
Brief Explanations
Ionic compounds form between metals (or polyatomic cations) and nonmetals (or polyatomic anions). Molecular compounds form between nonmetals (including metalloids in some cases).
- $\text{GaCl}_3$: Gallium (metal) + Chlorine (nonmetal) → Ionic
- $\text{ScN}$: Scandium (metal) + Nitrogen (nonmetal) → Ionic
- $\text{P}_4\text{O}_{10}$: Phosphorus (nonmetal) + Oxygen (nonmetal) → Molecular
- $\text{Na}_2\text{O}$: Sodium (metal) + Oxygen (nonmetal) → Ionic
- $\text{Bi}_2\text{S}_3$: Bismuth (metal) + Sulfur (nonmetal) → Ionic
- $\text{CdSe}$: Cadmium (metal) + Selenium (nonmetal) → Ionic
- $\text{CaBr}_2$: Calcium (metal) + Bromine (nonmetal) → Ionic
- $\text{SrI}$: Strontium (metal) + Iodine (nonmetal) → Ionic
- $\text{BH}_3$: Boron (metalloid) + Hydrogen (nonmetal) → Molecular
- $\text{CH}_3\text{OH}$: Carbon, Hydrogen, Oxygen (all nonmetals) → Molecular
- $\text{CO}_2$: Carbon (nonmetal) + Oxygen (nonmetal) → Molecular
- $\text{P}_2\text{O}$: Phosphorus (nonmetal) + Oxygen (nonmetal) → Molecular
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\text{GaCl}_3$: 2. Ionic
- $\text{ScN}$: 2. Ionic
- $\text{P}_4\text{O}_{10}$: 1. Molecular
- $\text{Na}_2\text{O}$: 2. Ionic
- $\text{Bi}_2\text{S}_3$: 2. Ionic
- $\text{CdSe}$: 2. Ionic
- $\text{CaBr}_2$: 2. Ionic
- $\text{SrI}$: 2. Ionic
- $\text{BH}_3$: 1. Molecular
- $\text{CH}_3\text{OH}$: 1. Molecular
- $\text{CO}_2$: 1. Molecular
- $\text{P}_2\text{O}$: 1. Molecular