QUESTION IMAGE
Question
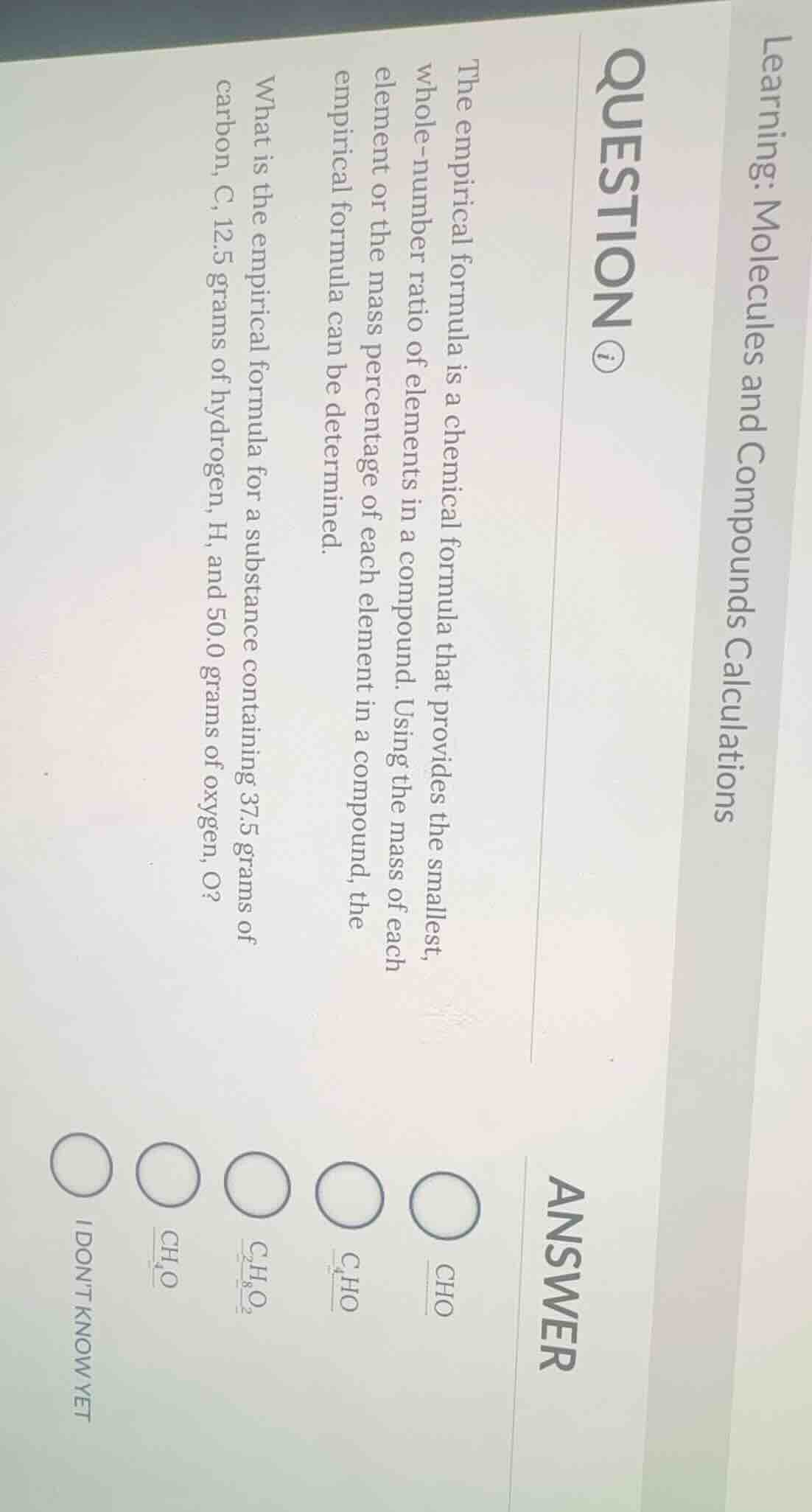
learning: molecules and compounds calculations
question ⓘ
the empirical formula is a chemical formula that provides the smallest, whole-number ratio of elements in a compound. using the mass of each element or the mass percentage of each element in a compound, the empirical formula can be determined.
what is the empirical formula for a substance containing 37.5 grams of carbon, c, 12.5 grams of hydrogen, h, and 50.0 grams of oxygen, o?
answer
○ cho
○ c₄ho
○ c₂h₈o₂
○ ch₄o
○ i dont know yet
Step1: Convert mass to moles
Molar mass of C = 12.01 g/mol, H = 1.008 g/mol, O = 16.00 g/mol
$n_C = \frac{37.5}{12.01} \approx 3.122$ mol
$n_H = \frac{12.5}{1.008} \approx 12.40$ mol
$n_O = \frac{50.0}{16.00} = 3.125$ mol
Step2: Find smallest mole value
Smallest $n \approx 3.122$ mol (from C)
Step3: Divide moles by smallest value
$Ratio_C = \frac{3.122}{3.122} = 1$
$Ratio_H = \frac{12.40}{3.122} \approx 4$
$Ratio_O = \frac{3.125}{3.122} \approx 1$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. $\text{CH}_4\text{O}$