QUESTION IMAGE
Question
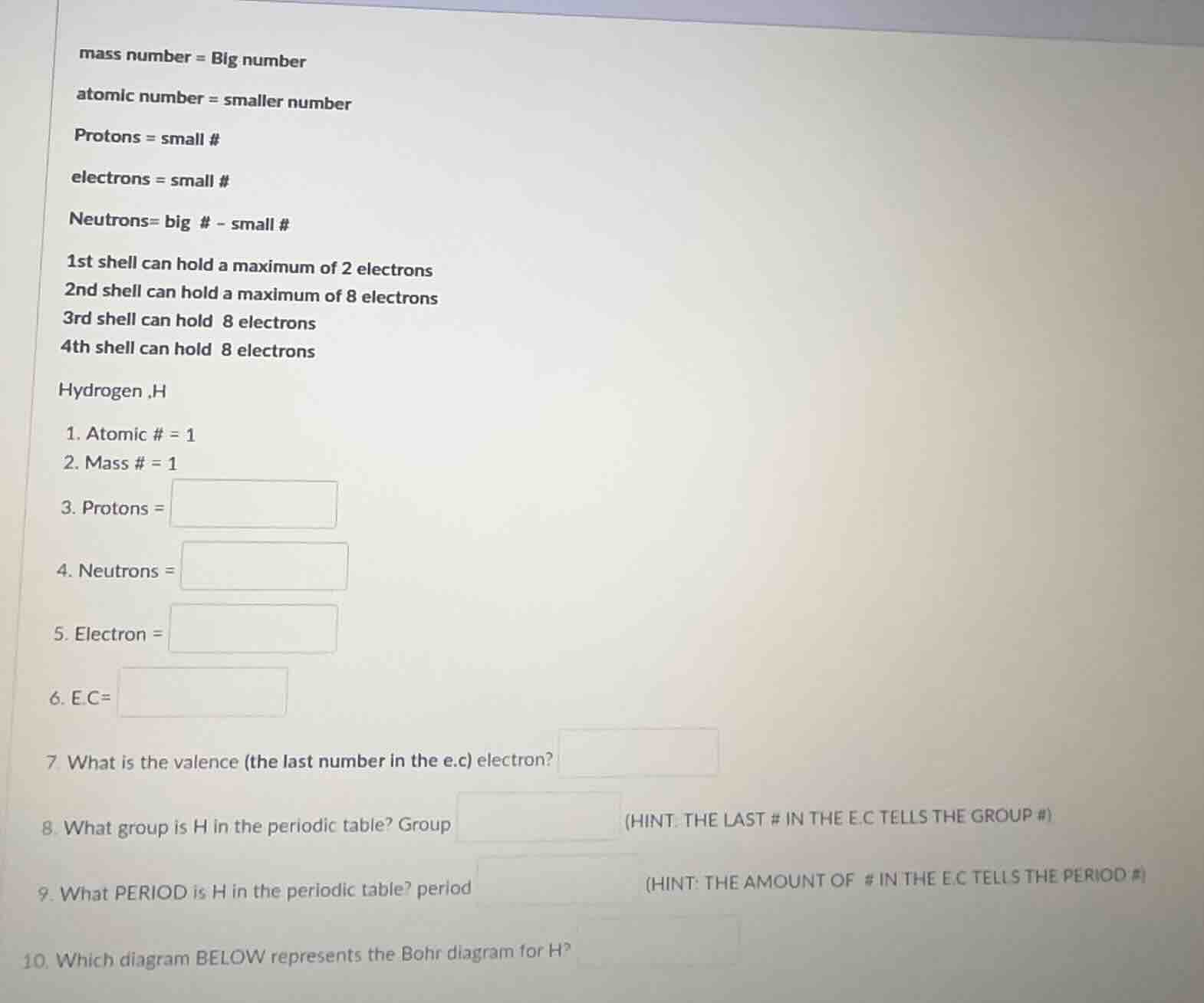
mass number = big number
atomic number = smaller number
protons = small #
electrons = small #
neutrons= big # - small #
1st shell can hold a maximum of 2 electrons
2nd shell can hold a maximum of 8 electrons
3rd shell can hold 8 electrons
4th shell can hold 8 electrons
hydrogen ,h
- atomic # = 1
- mass # = 1
- protons =
- neutrons =
- electron =
- e.c=
- what is the valence (the last number in the e.c) electron?
- what group is h in the periodic table? group (hint: the last # in the e.c tells the group #)
- what period is h in the periodic table? period (hint: the amount of # in the e.c tells the period #)
- which diagram below represents the bohr diagram for h?
Step1: Find protons (atomic #)
Protons = Atomic # = 1
Step2: Calculate neutrons
Neutrons = Mass # - Atomic # = $1 - 1 = 0$
Step3: Find electrons (atomic #)
Electrons = Atomic # = 1
Step4: Write electron configuration
1 electron fills 1st shell: $1$
Step5: Identify valence electron
Valence electron = last number in E.C. = 1
Step6: Find group number
Group # = valence electron count = 1
Step7: Find period number
Period # = number of shells used = 1
Step8: Describe Bohr diagram for H
Bohr diagram has 1 proton in nucleus, 1 electron in 1st (only) shell.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Protons = 1
- Neutrons = 0
- Electron = 1
- E.C= 1
- What is the valence (the last number in the e.c) electron? 1
- What group is H in the periodic table? Group 1
- What PERIOD is H in the periodic table? period 1
- Which diagram BELOW represents the Bohr diagram for H? A diagram with a central nucleus labeled "1 p⁺" (or just 1 proton) and one single electron orbiting in a single, outermost (and only) electron shell.