QUESTION IMAGE
Question
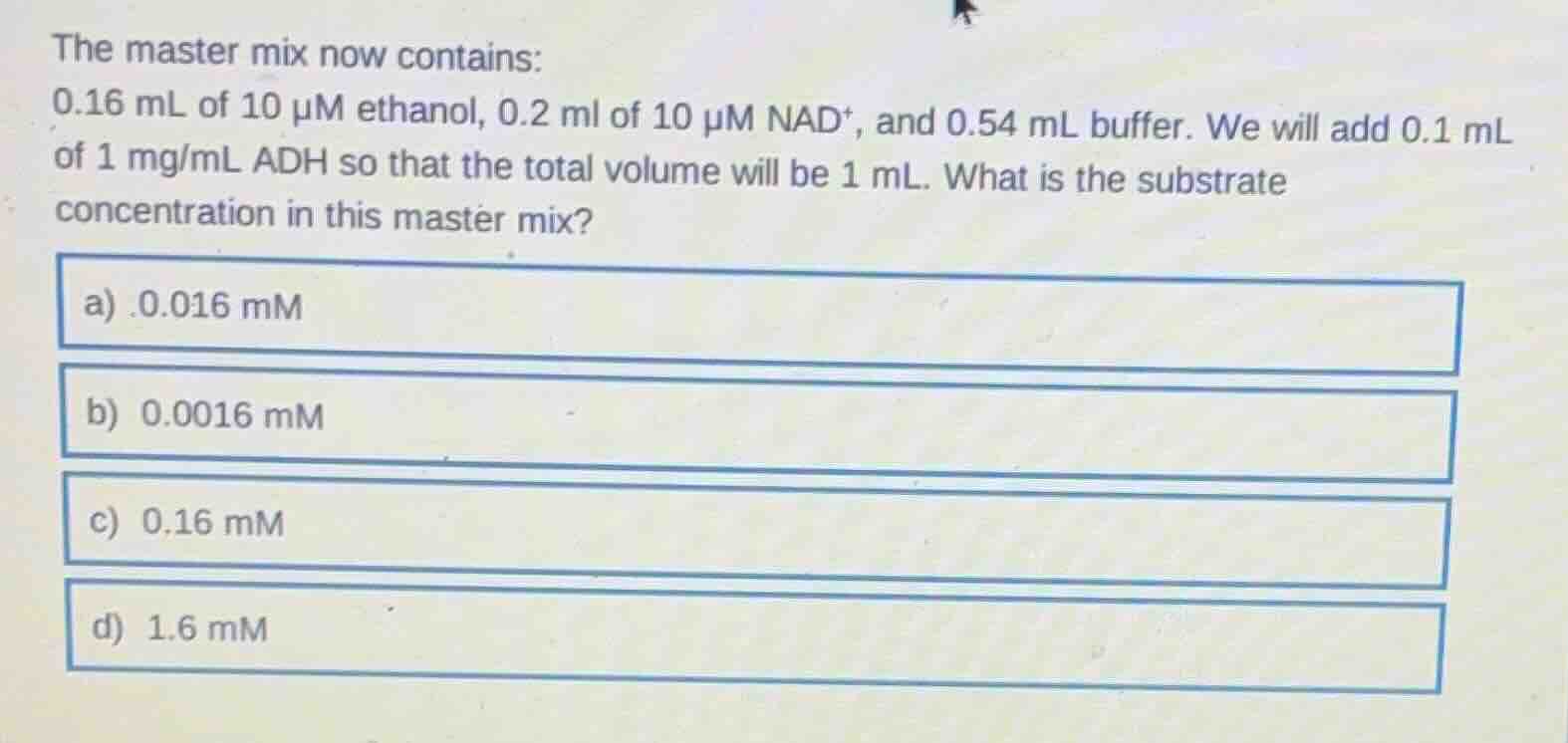
the master mix now contains:
0.16 ml of 10 μm ethanol, 0.2 ml of 10 μm nad⁺, and 0.54 ml buffer. we will add 0.1 ml of 1 mg/ml adh so that the total volume will be 1 ml. what is the substrate concentration in this master mix?
a) .0.016 mm
b) 0.0016 mm
c) 0.16 mm
d) 1.6 mm
Step1: Identify substrate and dilution law
The substrate is ethanol. Use dilution formula $C_1V_1 = C_2V_2$, where $C_1$ = initial concentration, $V_1$ = initial volume, $C_2$ = final concentration, $V_2$ = final total volume.
Step2: List known values
$C_1 = 10\ \mu\text{M}$, $V_1 = 0.16\ \text{mL}$, $V_2 = 1\ \text{mL}$
Step3: Solve for final concentration
Rearrange formula: $C_2 = \frac{C_1V_1}{V_2}$
Step4: Convert units to mM
Since $1\ \text{mM} = 1000\ \mu\text{M}$, $C_2 = \frac{1.6}{1000}\ \text{mM} = 0.0016\ \text{mM}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
b) 0.0016 mM