QUESTION IMAGE
Question
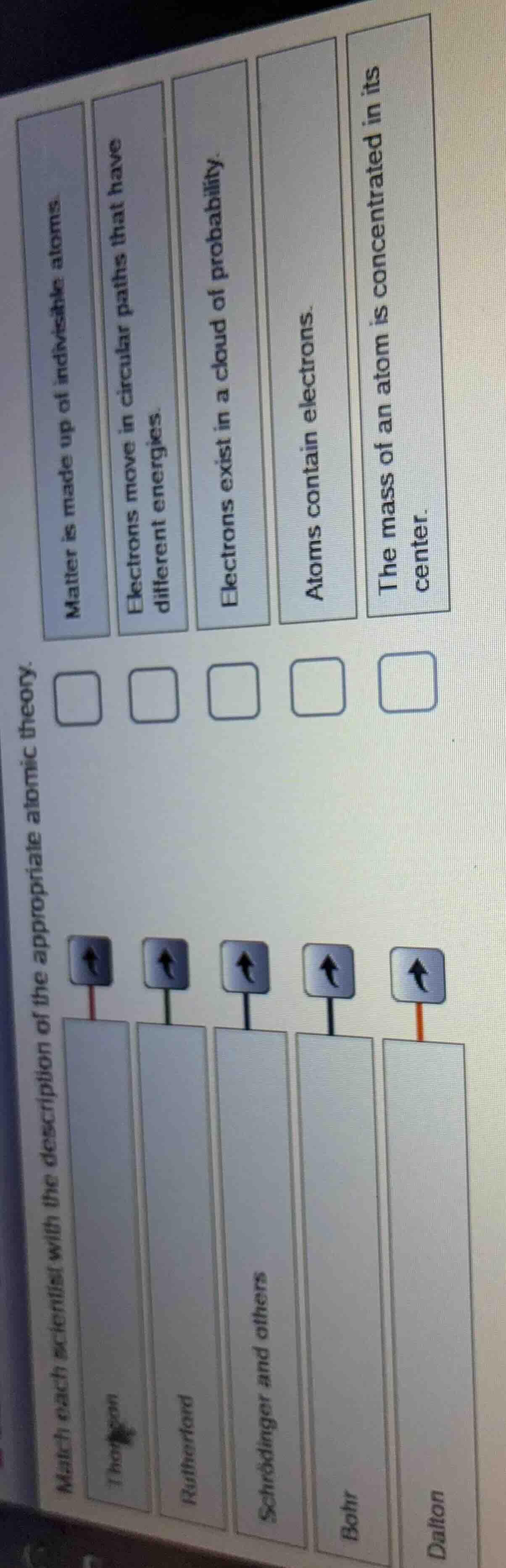
match each scientist with the description of the appropriate atomic theory.
thompson
rutherford
schrödinger and others
bohr
dalton
matter is made up of indivisible atoms.
electrons move in circular paths that have different energies.
electrons exist in a cloud of probability.
atoms contain electrons.
the mass of an atom is concentrated in its center.
Brief Explanations
Each match is based on the core claims of each scientist's atomic theory:
- Dalton proposed that all matter is composed of indivisible atoms.
- Bohr's model defined electrons moving in fixed circular energy levels.
- Schrödinger's quantum model describes electrons as a probability cloud.
- Thompson discovered the electron, proving atoms contain electrons.
- Rutherford's gold foil experiment showed most atomic mass is in a central nucleus.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Dalton: Matter is made up of indivisible atoms.
- Bohr: Electrons move in circular paths that have different energies.
- Schrödinger and others: Electrons exist in a cloud of probability.
- Thompson: Atoms contain electrons.
- Rutherford: The mass of an atom is concentrated in its center.