QUESTION IMAGE
Question
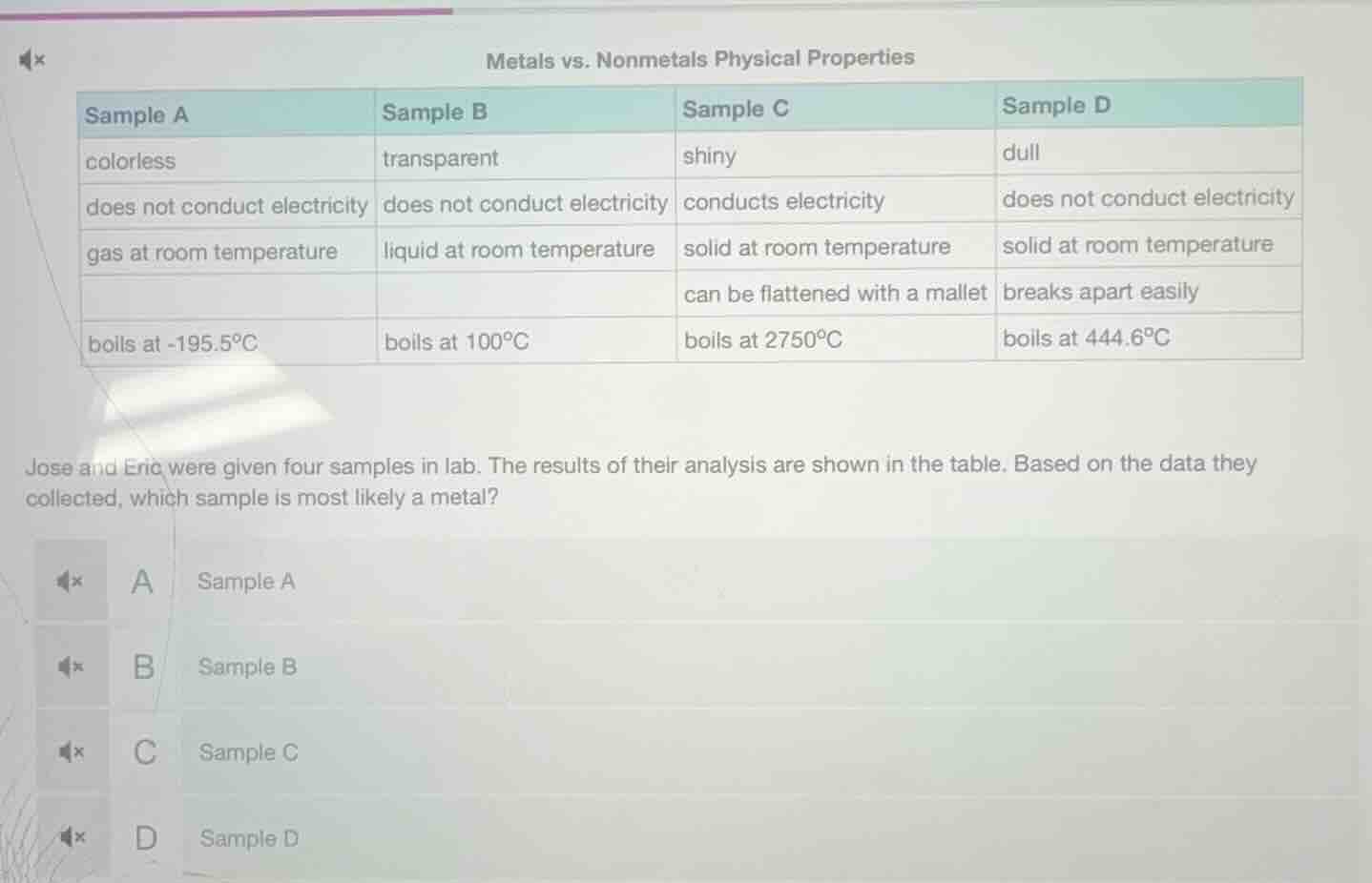
metals vs. nonmetals physical properties
sample a\tsample b\tsample c\tsample d
colorless\ttransparent\tshiny\tdull
does not conduct electricity\tdoes not conduct electricity\tconducts electricity\tdoes not conduct electricity
gas at room temperature\tliquid at room temperature\tsolid at room temperature\tsolid at room temperature
\t\tcan be flattened with a mallet\tbreaks apart easily
boils at -195.5°c\tboils at 100°c\tboils at 2750°c\tboils at 444.6°c
jose and eric were given four samples in lab. the results of their analysis are shown in the table. based on the data they collected, which sample is most likely a metal?
a sample a
b sample b
c sample c
d sample d
To determine which sample is a metal, we use typical metal properties: shiny, conducts electricity, solid at room temp, malleable (can be flattened), high boiling point.
- Sample A: gas, non - conductor → not metal.
- Sample B: liquid, non - conductor → not metal.
- Sample C: shiny, conducts electricity, solid, malleable, high boiling point → matches metal properties.
- Sample D: dull, non - conductor, brittle → not metal.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. Sample C